Retrospective Evaluation of Drug-Drug Interactions With Erlotinib and Gefitinib Use in the Military Health System
Background: Erlotinib and gefitinib are epidermal growth factor receptor–tyrosine kinase inhibitors approved for non–small cell lung cancer treatment by the US Food and Drug Administration. Drug-drug interactions (DDIs) with these agents are vague and poorly understood. Because DDIs can have an effect on clinical outcomes, we aimed to identify drugs that interact with erlotinib or gefitinib and describe their clinical manifestations.
Methods: A retrospective analysis was performed on the health records of patients in the US Department of Defense Cancer Registry (retrieved September 2021), Comprehensive Ambulatory/Professional Encounter Records, and Pharmacy Data Transaction Service database (both retrieved May 2022). Patients’ medical history, diagnoses, and demographics were extracted and analyzed for differences in adverse effects when these agents were used alone vs concomitantly with other prescription drugs. Patients’ diagnoses and prescription drug use were extracted to compare completed vs discontinued treatment groups, identify medications commonly co-administered with erlotinib or gefitinib, and evaluate DDIs with antidepressants.
Results: Of 387 patients using erlotinib, 264 completed treatments; 28 of 33 patients using gefitinib completed treatment. The P value for erlotinib discontinuation when used alone vs concomitantly was < .001, and the P value for gefitinib discontinuation was .06. Patients who took erlotinib or gefitinib concomitantly with a greater number of prescription drugs had a higher rate of treatment discontinuation than those who received fewer medications. Patients in the completed group received 1 to 75 prescription drugs, and those in the completed group were prescribed 3 to 103. Those who discontinued treatment had more diagnosed medical issues than those who completed treatment.
Conclusions: This review cannot conclude that concomitant use with prescription drug(s) resulted in erlotinib or gefitinib discontinuation. There were no significant DDIs determined between erlotinib or gefitinib and antidepressants.
Gefitinib
In September 2021, the Joint Pathology Center provided 33 entries for 33 patients who were systemically treated with gefitinib from January 1, 2002, to December 31, 2017. The patient ages ranged from 49 to 89 years with a mean age of 66 years. There were 31 (94%) and 2 (6%) patients with lung and other cancers, respectively. The upper lobe, lower lobe, and lung NOS had the most patients: 14 (42%), 8 (24%), and 6 (18%), respectively.
There were 31 patients (94%) who were aged > 50 years; 15 were male (45%) and 18 were female (55%). There were 26 patients (79%) who had a cancer diagnosis of stage III or IV disease. Nineteen patients (58%) were treated with gefitinib alone, and 14 (42%) were treated with gefitinib concomitantly with additional chemotherapy. Thirty-one patients (94%) were treated for lung cancer (Table 2). Thirty-three patients are a small sample size to determine whether patients were likely to stop gefitinib if used concomitantly with other drugs. Among the patients treated with gefitinib monotherapy, 5% (n = 1) stopped treatment, whereas 29% (n = 4) of patients treated concomitantly discontinued treatment (P = .06). All comparisons for gefitinib yielded insignificant P values. Physicians’ notes indicated that the reasons for gefitinib discontinuation were life-altering pruritis and unknown (progressive disease outcome) (Table 3).
Management Analysis and Reporting Tool Database
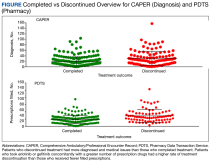
MHS data analysts provided data on diagnoses for 348 patients among 415 submitted, with 232 and 112 patients completing and discontinuing erlotinib or gefitinib treatment, respectively. Each patient had 1 to 104 (completed treatment group) and 1 to 157 (discontinued treatment group) unique health conditions documented. The MHS reported 1319 unique-diagnosis conditions for the completed group and 1266 for the discontinued group. Patients with additional health issues stopped chemotherapy use more often than those without; P < .001 for the completed group (232 patients, 1319 diagnoses) vs the discontinued group (112 patients, 1266 diagnoses). The mean (SD) number of diagnoses was 19 (17) for the completed and 30 (22) for the discontinued treatment groups (Figure).
MHS data was provided for patients who filled erlotinib (n = 240) or gefitinib (n = 18). Among the 258 patients, there were 179 and 79 patients in the completed and discontinued treatment groups, respectively. Each patient filled 1 to 75 (for the completed treatment group) and 3 to 103 (for the discontinued treatment group) prescription drugs. There were 805 unique-filled prescriptions for the completed and 670 for the discontinued group. Patients in the discontinued group filled more prescriptions than those who completed treatment; P < .001 for the completed group (179 patients,805 drugs) vs the discontinued group (79 patients, 670 drugs).
The mean (SD) number of filled prescription drugs was 19 (11) for the completed group and 29 (18) for the discontinued treatment group. The 5 most filled prescriptions with erlotinib from 258 patients with PDTS data were ondansetron (151 prescriptions, 10 recorded AEs), dexamethasone (119 prescriptions, 9 recorded AEs), prochlorperazine (105 prescriptions, 15 recorded AEs), oxycodone (99 prescriptions, 1 AE), and docusate (96 prescriptions, 7 recorded AEs).