Retrospective Evaluation of Drug-Drug Interactions With Erlotinib and Gefitinib Use in the Military Health System
Background: Erlotinib and gefitinib are epidermal growth factor receptor–tyrosine kinase inhibitors approved for non–small cell lung cancer treatment by the US Food and Drug Administration. Drug-drug interactions (DDIs) with these agents are vague and poorly understood. Because DDIs can have an effect on clinical outcomes, we aimed to identify drugs that interact with erlotinib or gefitinib and describe their clinical manifestations.
Methods: A retrospective analysis was performed on the health records of patients in the US Department of Defense Cancer Registry (retrieved September 2021), Comprehensive Ambulatory/Professional Encounter Records, and Pharmacy Data Transaction Service database (both retrieved May 2022). Patients’ medical history, diagnoses, and demographics were extracted and analyzed for differences in adverse effects when these agents were used alone vs concomitantly with other prescription drugs. Patients’ diagnoses and prescription drug use were extracted to compare completed vs discontinued treatment groups, identify medications commonly co-administered with erlotinib or gefitinib, and evaluate DDIs with antidepressants.
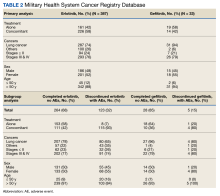
Results: Of 387 patients using erlotinib, 264 completed treatments; 28 of 33 patients using gefitinib completed treatment. The P value for erlotinib discontinuation when used alone vs concomitantly was < .001, and the P value for gefitinib discontinuation was .06. Patients who took erlotinib or gefitinib concomitantly with a greater number of prescription drugs had a higher rate of treatment discontinuation than those who received fewer medications. Patients in the completed group received 1 to 75 prescription drugs, and those in the completed group were prescribed 3 to 103. Those who discontinued treatment had more diagnosed medical issues than those who completed treatment.
Conclusions: This review cannot conclude that concomitant use with prescription drug(s) resulted in erlotinib or gefitinib discontinuation. There were no significant DDIs determined between erlotinib or gefitinib and antidepressants.
Results
Erlotinib
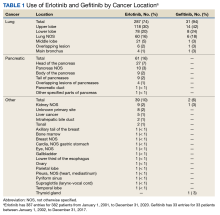
The Joint Pathology Center provided 387 entries for 382 patients aged 21 to 93 years (mean, 65 years) who were treated systemically with erlotinib from January 1, 2001, to December 31, 2020. Five patients had duplicate entries because they had different cancer sites. There were 287 patients (74%) with lung cancer, 61 (16%) with pancreatic cancer, and 39 (10%) with other cancers. For lung cancer, there were 118 patients (30%) for the upper lobe, 78 (20%) for the lower lobe, and 60 (16%) not otherwise specified (NOS). Other lung cancer sites had fewer patients: 21 (5%) middle lobe lung, 6 (2%) overlapping lung lesion(s), and 4 (1%) main bronchus of the lung. For pancreatic cancer, there were 27 patients (7%) for the head of the pancreas, 10 (3%) pancreas NOS, 9 (2%) body of the pancreas, 9 (2%) tail of the pancreas, 4 (1%) overlapping lesions of the pancreas, 1 (< 1%) pancreatic duct, and 1 (< 1%) other specified parts of the pancreas
There were 342 patients (88%) who were aged > 50 years; 186 male patients (48%) and 201 female patients (52%). There were 293 patients (76%) who had a cancer diagnosis of stage III or IV disease and 94 (24%) who had a cancer diagnosis of stage ≤ II (combination of data for stage 0, 1, and 2, not applicable, and unknown). For their systemic treatment, 161 patients (42%) were treated with erlotinib alone and 226 (58%) received erlotinib concomitantly with additional chemotherapy.
Patients were more likely to discontinue erlotinib for chemotherapy if they received concomitant treatment. Among the patients receiving erlotinib monotherapy, 5% stopped the treatment, whereas 51% of patients treated concomitantly discontinued (P < .001).
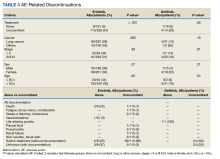
Among the 123 patients who discontinued their treatment, 101 switched treatment with no AEs notes, 22 died or experienced fatigue with blurry vision, constipation, nonspecific gastrointestinal effects, grade-4 diarrhea (as defined by the Common Terminology Criteria for Adverse Events), or developed a pleural fluid, pneumonitis, renal failure, skin swelling and facial rash, and unknown AEs of discontinuation. Patients who discontinued treatment because of unknown AEs had physicians’ notes that detailed emergency department visits, peripheral vascular disease, progressive disease, and treatment cessation, but did not specify the exact symptom(s) that led to discontinuation. The causes of death are unknown because they were not detailed in the available notes or databases. The overall results in this retrospective review cannot establish causality between taking erlotinib or gefitinib and death.