Analysis of Pharmacist Interventions Used to Resolve Safety Target of Polypharmacy (STOP) Drug Interactions
Background: Statin drug interactions commonly increase the risk of muscle-related toxicities. The medical literature supports consultative pharmacist interventions to resolve drug interactions, but studies demonstrating autonomous pharmacist interventions are lacking.
Objective: To evaluate the complementary impact of using pharmacist-led protocols and pharmacists with prescriptive authority to resolve statin drug interactions.
Methods: Pharmacist-led protocols were developed to address gemfibrozil-statin and niacin-statin interactions. Pharmacists discontinued gemfibrozil and niacin by protocol or referred patients to the Patient Aligned Care Team (PACT) Pharmacy Clinic for individualized management. After all drug interactions were addressed, a retrospective quality improvement analysis was conducted. The primary outcome was to evaluate the impact of gemfibrozil and niacin discontinuation by protocol on patients’ triglyceride (TG) laboratory results. The coprimary endpoints were the change in TGs and the percentage of patients with TGs ≥ 500 mg/dL, following pharmacist discontinuation by protocol. Secondary outcomes included the time required to resolve the interactions and a description of the PACT Clinical Pharmacy Specialists' (CPS) pharmacologic interventions.
Results: The gemfibrozil and niacin protocols addressed 397 drug interactions. Seventy-six patients underwent gemfibrozil discontinuation by protocol and had TG laboratory results available. TG levels decreased or increased by < 100 mg/dL for 62 patients (82%), and 1 patient (1.3%) experienced TG elevation above the threshold of 500 mg/dL. Thirty-six patients had niacin discontinued by protocol and available laboratory results. The TG levels decreased or increased by < 100 mg/dL for 33 patients (91.7%), and no patients had TG levels increase above the threshold of 500 mg/dL. The mean time required to resolve both gemfibrozil and niacin drug interactions was 15.5 minutes per patient. A total of 129 patients were referred to the PACT Pharmacy Clinic to manage gemfibrozil and niacin drug interactions. TG laboratory results were available for 80 gemfibrozil patients (74.8%) and 16 niacin patients (72.7%). The PACT CPS made 171 pharmacologic interventions to address drug interactions and the median of 2 encounters per patient.
Conclusions: This single-site quality improvement analysis supports the complementary use of protocols and pharmacists with prescriptive authority to resolve statin drug interactions. These data support expanded roles for pharmacists, across settings, to mitigate select drug interactions.
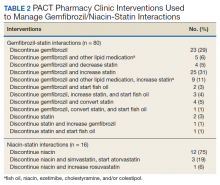
Of the 107 patients referred to the PACT Pharmacy Clinic, 80 (74.8%) had TG laboratory results available and were included in the analysis. These patients were followed by the PACT CPS until the drug interaction was resolved and confirmed to have TG levels at goal (< 500 mg/dL). Gemfibrozil doses ranged from 300 mg daily to 600 mg twice daily, with 70% (n = 56) of patients taking 600 mg twice daily. The PACT CPS made 148 interventions (Table 2). Twenty-three (29%) patients required only gemfibrozil discontinuation. The remaining 57 patients (71%) required at least 2 medication interventions. The PACT CPS generated 213 encounters for resolving drug interactions with a median of 2 encounters per patient.
Quality assurance review identified 5 patients (5.3%) who underwent gemfibrozil discontinuation by protocol, despite having criteria that would have recommended against discontinuation. In accordance with the protocol criteria, these patients were later referred to the PACT Pharmacy Clinic. None of these patients experienced a TG increase at or above the threshold of 500 mg/dL after gemfibrozil was initially discontinued but were excluded from the earlier analysis.
Niacin-Statin Interactions
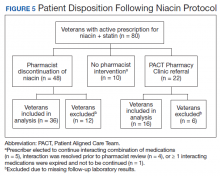
Pharmacists discontinued niacin by protocol for 48 patients (60.0%), and 22 patients (27.5%) were referred to the PACT Pharmacy Clinic (Figure 5). For the remaining 5 patients (6.3%), the interaction was either addressed outside the protocol prior to pharmacist review, or an interacting prescription was expired and not to be continued. Additionally, niacin was continued per prescriber preference in 5 patients (6.3%).
Thirty-six patients (75%) had TG laboratory results available following niacin discontinuation by protocol and were included in the analysis. Most patients’ (n = 33, 91.7%) TG levels decreased or increased by < 100 mg/dL. No patient had a TG level that increased higher than the threshold of 500 mg/dL. The mean (SD) time to the first laboratory result after the pharmacists mailed the notification letter, was 5.3 (2.5) months (range, 1.2-9.8). The pharmacists spent a mean of 15 minutes per patient resolving each interaction. The quality assurance review found no discrepancies in the pharmacists’ application of the protocol.
Of the 22 patients referred to the PACT Pharmacy Clinic, 16 (72.7%) patients had TG laboratory results available and were included in the analysis. As with the gemfibrozil interactions, these patients were followed by the PACT Pharmacy Clinic until the drug interaction was resolved and confirmed to have TGs at goal (< 500 mg/dL). Niacin doses ranged from 500 mg daily to 2,000 mg daily, with the majority of patients taking 1,000 mg daily. The PACT CPS made 23 interventions. The PACT CPS generated 46 encounters for resolving drug interactions with a median of 2 encounters per patient.