Aromatase inhibitors, a new option for inducing ovulation
This class of drugs may boost the pregnancy rate in selected populations
IN THIS ARTICLE
Third-generation AIs that are commercially available in North America, Europe, and other parts of the world include:
- two nonsteroidal preparations: anastrozole (Arimidex) and letrozole (Femara)
- one steroidal agent: exemestane (Aromasin).
Letrozole and anastrozole are reversible, competitive agents with considerably greater potency (more than 1,000 times greater) than the first-generation AI aminoglutethimide. At a dosage of 1 to 5 mg/day, they reduce estrogen levels by 97% to more than 99%.
AIs are completely absorbed after oral administration, with a mean terminal half-life of approximately 45 hours (range: 30–60 hours). Exemestane has a shorter circulating half-life of approximately 9 hours, but may have a longer effect because it is irreversible.21
Mild gastrointestinal (GI) disturbances account for most of the adverse events, and rarely limit therapy.
How AIs work
Although we continue to accrue data on the use of AIs to induce ovulation, the underlying mechanism of action has not been studied. However, we believe that AIs work both centrally (at the level of the hypothalamus and pituitary) and peripherally (at the level of the ovaries).22-28
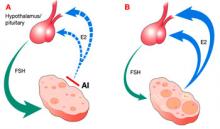
At the central level, AIs suppress estrogen production by directly, specifically, and potently inhibiting the aromatase enzyme (i.e., estrogen synthase, the enzyme responsible for the synthesis of estrogen). Because the aromatase enzyme is expressed in various tissues and organs—most notably, the ovaries, brain, and fat29—AIs suppress estrogen production in all of those tissues, leading to a low serum estrogen level and low local estrogen level. Low estrogen levels are thought to release the hypothalamus and pituitary gland from their negative-feedback mechanism, thereby increasing production of endogenous gonadotropins from the pituitary gland and stimulating ovarian follicular development and ovulation (FIGURE).
At the peripheral level, the aromatase enzyme catalyzes the terminal step in the steroidogenesis cascade that converts androgens into estrogen. When that enzyme is inhibited, enzyme substrate (androgens) is thought to accumulate. Contrary to the general belief that androgens are deleterious to ovarian follicles, studies in primates have demonstrated that androgens actually up-regulate the expression of gonadotropin receptors, particularly follicle-stimulating hormone (FSH) receptors.30 This renders the ovaries more sensitive to gonadotropin stimulation—whether the gonadotropins are endogenous or exogenous.22-28
FIGURE Aromatase inhibitors promote follicle development, then fade from the scene in time to prevent hyperovulation
Administration of an aromatase inhibitor (AI) on cycle days 3 to 7 suppresses ovarian estradiol (E2) secretion, as shown in A, which reduces estrogen-negative feedback at the hypothalamus and pituitary. As a result, follicle-stimulating hormone (FSH) secretion increases, fostering growth of multiple ovarian follicles. The growing follicles, shown in B, cause estrogen levels to rise again, depressing FSH, and leading to monofollicular ovulation in most cases.
Why AIs are superior to clomiphene
Clomiphene citrate is a selective estrogen receptor modulator (SERM) that is believed to induce ovulation through its antiestrogenic properties at the level of the hypothalamus or pituitary gland, or both. Clomiphene down-regulates estrogen receptors at this level, and the hypothalamus and pituitary gland react as though the estrogen level is very low. This reverses the suppression of endogenous gonadotropins by estrogen, and gonadotropin levels rise, stimulating ovarian follicular development.
The down-regulation of estrogen receptors with clomiphene administration is not limited to the hypothalamus and pituitary gland, but also occurs peripherally at the endometrium and cervix, where it is not so desirable. When the cervix is affected, it becomes an unfavorable environment for sperm to penetrate, and when the endometrium is affected, its hypoestrogenic status may reduce the likelihood of embryo implantation—or may increase the risk of pregnancy loss if implantation occurs.
These peripheral antiestrogenic prop erties of clomiphene citrate may account for the discrepancy between high ovulation and low pregnancy rates.22-28 Several strategies to overcome this problem—e.g., adding estrogen, starting clomiphene citrate earlier in the menstrual period, or using another SERM, such as tamoxifen—have been largely unsuccessful. With clomiphene citrate, depletion of estrogen receptors has long-term effects because of the drug’s relatively long half-life (several days).31
In contrast, AIs do not appear to affect the expression of estrogen receptors in different body tissues, such as the endometrium and cervix. AIs have a shorter half-life (8 hours to 2 days), and nonsteroidal third-generation agents have a reversible inhibitory effect on the aromatase enzyme. Moreover, the rise in endogenous gonadotropins stimulates the production of more aromatase enzyme. This newly formed aromatase enzyme, and the return of a normal aromatase level after a short half-life of AI, leads the maturing ovarian follicles to secrete estrogen, which reaches a physiologic level soon after the last administration of AI. The rising estrogen level allows development of a more hospitable uterine environment (endometrium and cervical mucus).22-28