The VA Research Enterprise: A Platform for National Partnerships Toward Evidence Building and Scientific Innovation
Background: Within a year of the start of the COVID-19 pandemic, the US Department of Veterans Affairs (VA) was managing about 300 COVID-19–related research projects across roughly 100 facilities, which has since grown to more than 900 projects. This robust set of activities arose from an existing enterprise strategy and aimed at identifying needs for supporting the clinical care mission, more rapidly leveraging resources, and coordinating research across the VA. The VA’s efforts to implement an enterprise strategy before March 2020 positioned its research community to dynamically partner with other federal agencies, academic institutions, and industry in addressing a national public health emergency.
Observations: The VA research enterprise involves a broad range of functions, scientific and clinical leaders, and organizational resources to enhance the health and care of veterans and the nation. The scope of research activities enables it to support its priorities while also partnering with others who share in mutual commitments to veteran health. Moving toward being the nation’s learning health care system, the VA’s leadership support, staff, patient volunteers, and partners were key contributors to a national response to COVID-19. Swift action and consistent communication helped address the complexities of the pandemic and strengthened the VA’s ability to prepare and mobilize for emergencies and other potential disease outbreaks. Documenting strategies and practices can enhance future opportunities aimed at addressing the most challenging health care needs while also focusing on the primary mission to serve veterans.
Conclusions: The COVID-19 pandemic contributed to critical knowledge and lessons that enabled the VA to advance enterprise goals, particularly in the context of its health care system. Sharing these unique processes and experiences will inform current and future partnerships among research, clinical, and public health communities oriented to serve veterans and the nation through scientific innovation.
ORGANIZING THE VA COVID-19 RESEARCH RESPONSE
VA Research seeks to internally standardize and integrate collaborations with clinical and operational partners throughout the agency. When possible, it seeks to streamline partnership efforts involving external groups less familiar with how the VA operates or its policies, as well as its capabilities. This need was more obvious during the pandemic, and the ORD assembled its COVID-19 response quickly.6
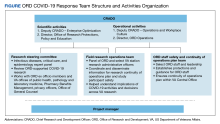
In early January 2020, VA offices, including the ORD, were carefully observing COVID-19. On March 4, 2020, a week before the World Health Organization declared COVID-19 a pandemic, the ORD and its National Research Advisory Council arranged a briefing from VA public health leaders to deal with reported cases of COVID-19 and VA plans. Immediately afterward, the ORD Chief Research and Development Officer gathered a team of experts in clinical research, infectious disease, and public health to strategize a broader research enterprise approach to the pandemic. This group quickly framed 3 key targets: (1) identify critical research questions to prioritize; (2) provide operational guidance to the research community; and (3) uphold VA research staff safety. This discussion led to the creation of a larger ORD COVID-19 Research Response Team that managed activities within this scope. This team included other ORD leaders and staff with operational, scientific, and regulatory expertise charged with enterprise-level planning and execution for all research activities addressing or affected by the pandemic (Figure).
Effective and timely communication was chief among key ORD responsibilities. On March 19, 2020, the Response Team informed the VA Research community about ORD plans for organizing the VA COVID-19 research response.7 It also mobilized VA research programs and investigators to support an enterprise approach that would be coordinated centrally. We achieved communication goals by developing a dedicated website, which provided a means to distribute up-to-date notices and guidance, answer frequently asked questions, and alert investigators about research opportunities. The site enabled the field to report on its efforts, which enhanced leadership and community awareness. A working group of ORD and field personnel managed communications. Given the volume of existing non–COVID-19 research, we established a research continuity of operations plan to provide guidelines for study participant and research staff safety. The ORD issued an unprecedented full-stop administrative hold on in-person research activities after the global announcement of the pandemic. This policy provided formal protections for research programs to safeguard staff and research participants and to determine appropriate alternatives to conduct research activities within necessary social distancing, safety, and other clinical care parameters. It also aligned with guidance and requirements that local VAMCs issued for their operations and care priorities.
The Response Team also established a scientific steering committee of VA infectious disease, critical care, informatics, and epidemiology experts to prioritize research questions, identify research opportunities, and evaluate proposals using a modified expeditious scientific review process. This group also minimized duplicate scientific efforts that might be expected from a large pool of investigators simultaneously pursuing similar research questions. Committee recommendations set up a portfolio that included basic science efforts in diagnostics, clinical trials, population studies, and research infrastructure.