Using Active Surveillance to Identify Monoclonal Antibody Candidates Among COVID-19–Positive Veterans in the Atlanta VA Health Care System
Background: Strategies for optimizing identification and outreach to potential candidates for monoclonal antibody (Mab) therapy for COVID-19 are not clear. Using a centralized, active surveillance system, the Atlanta Veterans Affairs Health Care System (AVAHCS) infectious disease (ID) team identified candidates for Mab infusion and provided treatment.
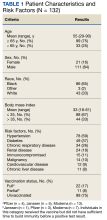
Observations: As part of a quality improvement project from December 28, 2020, to August 31, 2021, a clinical team consisting of ID pharmacists and physicians reviewed each outpatient with a positive COVID-19 polymerase chain reaction test daily at the AVAHCS. The clinical team used Emergency Use Authorization (EUA) criteria to determine eligibility. Eligible patients were contacted on the same day of review via telephone to confirm eligibility and obtain verbal consent. Telehealth follow-up occurred on day 1 and day 7 postinfusion to assess for adverse events. In total, 2028 patients with COVID-19 were identified; 289 patients (14%) were eligible, and 132 (46%) received Mab therapy. Similar to AVAHCS demographics, a majority of those who received Mab therapy were non-Hispanic Black patients (65%). The most common comorbidities were hypertension (59%) and diabetes (37%). The median time from symptom onset to positive COVID-19 polymerase chain reaction (PCR) test result was 6 days (range, 0-9), and the median time from positive COVID-19 PCR test result to Mab infusion was 2 days (range, 0-8). Twelve patients (9%) required hospitalization for worsening COVID-19 symptoms postinfusion. No deaths occurred.
Conclusions: Combining laboratory surveillance and active screening led to high uptake of Mab therapy and minimized delay from symptom onset to Mab infusion, thereby optimizing outpatient treatment of COVID-19. This approach also successfully screened and treated Black patients in the AVAHCS population.
Implementation
AVAHCS serves a diverse population of more than 129,000 (50.8% non-Hispanic Black veterans, 37.5% White veterans, and 11.7% of other races) at a main medical campus and 18 surrounding community-based outpatient clinics. From December 28, 2020, to August 31, 2021, veterans with a positive COVID-19 nasopharyngeal polymerase chain reaction (PCR) test at AVAHCS were screened daily. A central Mab team consisting of infectious disease (ID) clinical pharmacists and physicians reviewed daily lists of positive laboratory results and identified high-risk individuals for Mab eligibility, using the FDA EUA inclusion criteria. Eligible patients were called by a Mab team member to discuss Mab treatment, provide anticipatory guidance, obtain verbal consent, and schedule the infusion. Conventional referrals from non-Mab team members (eg, primary care physicians) were also accepted into the screening process and underwent the same procedures and risk prioritization strategy as those identified by the Mab team.
Clinic resources allowed for 1 to 2 patients per day to be given Mab, increasing to a maximum of 5 patients per day during the COVID-19 Delta variant surge. We followed our best clinical judgment in prioritizing patient selection, and we aligned our practice with the standards of our affiliated partner, Emory University. In circumstances where patients who were Mab-eligible outnumbered infusion availability, patients were prioritized using the Veterans Health Administration (VHA) COVID-19 (VACO) Index for 30-day COVID-19 mortality.8 As COVID-19 variants developed resistance to the recommended Mab infusions, bamlanivimab, bamlanivimab-etesevimab, or casirivimab-imdevimab, local protocols adapted to EUA revisions. The Mab team also adopted FDA eligibility criteria revisions as they were available.9,10
We describe the outcomes of our centralized screening process for Mab therapy, as measured by screening, uptake, and time to receipt of Mab from screening. We also describe the demographic and clinical characteristics of Mab recipients. Clinical outcomes include postinfusion adverse events (AEs) at day 1 and day 7, emergency department (ED) visits, inpatient hospitalization, and death.
Results
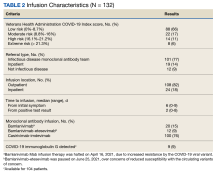
The Mab team screened 2028 veterans who were COVID-19 positive between December 28, 2020, and August 31, 2021, and identified 289 veterans (14%) who met the EUA criteria. One hundred thirty-two veterans (46%) completed Mab infusion, and of the remaining 145 veterans, 124 (86%) declined treatment, and 21 (14%) veterans did not complete Mab infusion largely due to not keeping the appointment. The Mab team active surveillance strategy identified 101 of 132 infusion candidates (77%); 82% had outpatient Mab infusion.
The mean age of veterans who received Mab was 55 years (range, 29-90), and 75% of veterans were aged ≥ 65 years; most were male (84%) and 86 (65%) identified as non-Hispanic Black individuals (Table 1).
Postinfusion AEs reported at day 1 and day 7 occurred for 38 veterans (29%) and 11 veterans (8%), respectively. Sixteen patients (12%) had postinfusion ED visit, and 12 patients (9%) required hospitalization. Eleven of the 12 hospitalized patients (92%) had worsening respiratory symptoms. No deaths occurred in the 132 patients who received Mab.