Evaluation of Gabapentin and Baclofen Combination for Inpatient Management of Alcohol Withdrawal Syndrome
Background: Benzodiazepines are considered the gold standard for treatment of alcohol withdrawal syndrome (AWS), a group of symptoms that occur after abrupt cessation of alcohol use, but may be associated with serious adverse effects. Given the safety concerns, alternative treatment options for AWS management have been investigated, including gabapentin and baclofen. Because no available studies have investigated the inpatient use of the gabapentin and baclofen combination for alcohol detoxification, this study aims to evaluate their efficacy and safety in the inpatient hospital setting.
Methods: This retrospective cohort study at the Captain James A. Lovell Federal Health Care Center in North Chicago, Illinois, included patients who were aged ≥ 18 years and who were admitted to the general acute medicine floor for the primary indication of AWS from January 1, 2014, to July 31, 2021. The primary outcome was the length of stay, defined as hours from admission to either discharge or 36 hours with a Clinical Institute Withdrawal Assessment of Alcohol (CIWA) score ≤ 8. Electronic health records were reviewed to collect CIWA scores, alcohol withdrawal seizure and delirium tremens incidence, rates of conversions from gabapentin/baclofen to lorazepam, rates of transitions to a higher level of care, and readmission for AWS within 30 days.
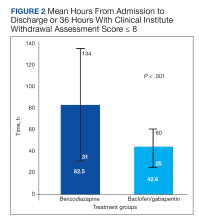
Results: Mean length of stay in the gabapentin/baclofen group was statistically significantly shorter compared with the benzodiazepine group (42.6 vs 82.5 hours, P < .001). The study found no significant difference between the gabapentin/baclofen and benzodiazepine groups in AWS readmission, adjuvant medications for AWS management, and number of patients who transitioned to a higher level of care. Overall, the safety of gabapentin/baclofen vs benzodiazepine were comparable; however, 1 patient experienced a seizure, and 1 patient experienced delirium tremens during admission in the benzodiazepine group.
Conclusions: Gabapentin/baclofen combination seems to be an effective and safe alternative to benzodiazepines and may be considered for managing mild AWS in hospitalized patients, but additional research is needed to examine this regimen.
The primary outcome of this study was the length of stay (LOS), which was
CPRS was used to collect information including baseline demographics, blood alcohol content, CIWA scores throughout hospitalization, number of admissions for alcohol detoxification in the previous year, AWS readmission within 30 days after discharge, prior treatment with g/b, history of alcohol withdrawal seizures and DTs, hospital LOS, outpatient medications for AUD treatment, rates of conversions from g/b protocol to lorazepam, and rates of transition to a higher level of care.
Statistical Analysis
Study data were stored and analyzed using an Excel spreadsheet and IBM SPSS Statistics software. LOS was compared between the g/b and benzodiazepine groups using inferential statistics. An independent 2-sample t test was used to assess the primary outcome if data were normally distributed. If the collected data were not distributed normally, the Mann-Whitney U test was used. All other continuous variables were assessed by using independent t tests and categorical variables by using χ2 tests. A P value < .05 was considered statistically significant. Effect size of d = 0.42 was calculated based on a previous study with a similar research design as our study.9 We determined that if using an independent 2-sample t test for the primary outcome analysis, an estimated sample size of 178 subjects would provide the study with an 80% power to detect a difference at a 2-sided significance level with α = 0.05. If using the Mann-Whitney U test, 186 subjects would be required to provide identical power.
Results
We reviewed 196 patient health records, and 39 were initially excluded. The most common reason was that AWS was not the primary diagnosis for hospitalization (n = 28).
The Shapiro-Wilk tests showed a significant departure from normality in the benzodiazepine group W(35) = 0.805 (P < .001) and g/b group W(20) = 0.348 (P < .001) for the primary outcome.
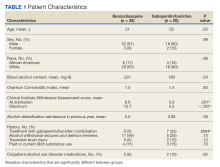
Additionally, this study examined multiple secondary outcomes (Table 2).