Cardiac Adverse Events Following COVID-19 Vaccination in Patients With Prior Vaccine-Associated Myocarditis
Background: Limited information exists to guide shared clinical decision making on COVID-19 vaccination in persons with a prior history of vaccine-associated myocarditis, pericarditis, or myopericarditis (VAMP). The objective of this retrospective observational case series was to characterize cardiac outcomes within 30 days following receipt of 1 or more COVID-19 vaccinations during 2021 in US service members diagnosed with prior non-COVID-19 VAMP between 1998 and 2019.
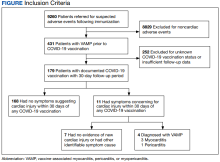
Methods: As part of the collaborative public health mission with the Centers for Disease Control and Prevention for enhanced vaccine adverse events surveillance, the Defense Health Agency Immunization Healthcare Division maintains a clinical database of service members and beneficiaries referred for suspected adverse events following immunizations. Cases in this database recorded between January 1, 2003, and February 28, 2022, were reviewed to identify individuals with prior VAMP who received a COVID-19 vaccine in 2021 and developed signs or symptoms suggestive of VAMP within 30 days following COVID-19 vaccination.
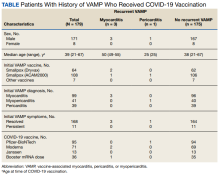
Results: Before the COVID-19 pandemic, 431 service members had verified VAMP. Among these 431 patients, 179 had records that confirmed receipt of a COVID-19 vaccine in 2021. Of these 179 patients, 171 (95.5%) were male. Their median age was 39 years (range, 21-67) at the time of COVID-19 vaccination. Most (n = 172; 96.1%) experienced their original VAMP episode after receipt of the live replicating smallpox vaccine. Eleven patients experienced cardiac-suggestive symptoms (chest pain, palpitations, or dyspnea) within 30 days of COVID-19 vaccination. Four patients met the criteria for recurrent VAMP. Three men aged 49, 50, and 55 years developed myocarditis within 3 days of an mRNA COVID-19 vaccine. One 25-year-old man developed pericarditis within 4 days of receiving an mRNA vaccine. All 4 COVID-19 recurrent VAMP cases fully recovered with minimal supportive care within weeks (myocarditis) to months (pericarditis).
Conclusions: As demonstrated by this case series, albeit rare, VAMP may reoccur after COVID-19 vaccination among patients who experienced cardiac injury after smallpox vaccination. The clinical characteristics and course of the 4 recurring cases were mild, appearing similar to the post-COVID-19 VAMP described in individuals without a history of VAMP. More research is warranted on factors that may predispose patients to vaccine-associated cardiac injury and which vaccine platforms or schedules may reduce the risk of recurrence among patients who have experienced these events.
Vaccinations have substantially reduced morbidity and mortality from many infectious diseases. Despite the clear value of vaccinations in public health, efforts to better understand adverse events (AEs) following immunization are important to sustain public trust and vaccine confidence. Noninfectious inflammation of the heart may manifest as myocarditis or pericarditis, or occasionally, with shared signs and symptoms of each, as myopericarditis. This is a rare AE following some immunizations. Vaccine-associated myocarditis, pericarditis, or myopericarditis (VAMP) has been most clearly associated with smallpox vaccines and mRNA COVID-19 vaccines.1-6 Although extremely rare, VAMP also has been associated with other vaccines.7,8 Limited information exists to guide shared clinical decision making on COVID-19 vaccination in persons with a history of VAMP. It is unknown whether individuals with a history of VAMP are at higher risk for developing a recurrence or experiencing a more severe outcome following COVID-19 vaccination.
Methods
As part of the collaborative public health mission with the Centers for Disease Control and Prevention (CDC) for enhanced vaccine AE surveillance, the Defense Health Agency Immunization Healthcare Division (IHD) maintains a clinical database of service members and beneficiaries referred for suspected AEs following immunizations. A review of all AEs following immunization cases in this database from January 1, 2003, through February 28, 2022, identified individuals meeting the following criteria: (a) VAMP prior to receipt of COVID-19 vaccine; (b) receipt of COVID-19 vaccine in 2021; and (c) medical documentation in available electronic health records sufficient to describe health status at least 30 days following COVID-19 vaccination.9 If medical entries suggested cardiac symptoms following a COVID-19 vaccine, additional information was sought to verify VAMP based on current published criteria.10,11 Both the initial VAMP cases and the suspected COVID-19 VAMP cases were adjudicated by a team of vaccine experts and specialists in immunology, cardiology, and preventive medicine.
This retrospective review was approved and conducted in accordance with the Walter Reed National Military Medical Center Institutional Review Board protocol #20664. All individuals with recurrent VAMP consented to share their health records and clinical details.
Results
Among 9260 cases in the IHD database, 431 met the case definition for VAMP.
Among the 179 patients included in this analysis, 171 (96%) were male. Their median age was 39 years at the time of COVID-19 vaccination.