The Safety and Efficacy of AUC/MIC-Guided vs Trough-Guided Vancomycin Monitoring Among Veterans
Background: Vancomycin is a commonly used antibiotic for the treatment of methicillin-resistant Staphylococcus aureu s (MRSA), which requires therapeutic drug monitoring (TDM). Guidelines recommend targeting an individualized area under the curve/minimum inhibitory concentration (AUC/MIC) ratio of 400 to 600 mg × h/L to maximize efficacy and minimize the risk of acute kidney injury (AKI). Before these guidelines, the accepted method of vancomycin TDM was using trough levels alone. To our knowledge, no studies of veterans have compared the difference in AKI incidence and time in the therapeutic range between monitoring strategies.
Methods: This single-site, retrospective, quasi-experimen tal study was conducted at the Sioux Falls Veterans Affairs Health Care System. The primary endpoint was the difference in vancomycin-induced AKI incidence between the 2 groups.
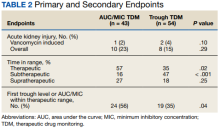
Results: This study included 97 patients with 43 in the AUC/MIC group and 54 in the trough-guided group. The incidence of vancomycin-induced AKI was 2% in the AUC/MIC group and 4% in the trough group ( P = .10). The incidence of overall AKI for AUC/MIC-guided and trough-guided TDM was 23% and 15% ( P = .29), respectively.
Conclusions: We did not find a significant difference in the incidence of vancomycin-induced or overall AKI between AUC/MIC- and trough-guided TDM. However, this study did indicate that AUC/MIC-guided TDM of vancomycin may be more effective than trough-guided TDM regarding a quicker time to and higher overall time in the therapeutic range. These findings support the recommendation to transition to the use of AUC/MIC-guided TDM of vancomycin in the veteran population.
For AUC/MIC-guided TDM the Cmax levels were ideally drawn 1 to 2 hours after vancomycin infusion and Cmin levels were ideally drawn 30 minutes before the next dose. First-order pharmacokinetic equations were used to estimate the AUC/MIC.12 If the timing of a vancomycin level was inappropriate, actual levels were extrapolated based on the timing of the blood draw compared with the ideal Cmin or Cmax time. Extrapolated levels were used for both trough-guided and AUC/MIC-guided TDM groups when appropriate. Vancomycin levels were excluded if they were drawn during the vancomycin infusion.
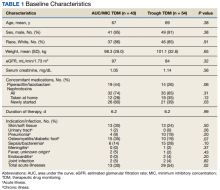
Study participant age, sex, race, weight, baseline estimated glomerular filtration (eGFR) rate, baseline sCr, concomitant nephrotoxic medications, duration of vancomycin course, indication of vancomycin, and acuity of illness based on indication were collected. sCr levels were collected from the initial day vancomycin was ordered through 72 hours following completion of a vancomycin course to evaluate for AKI. Patients’ charts were reviewed for the use of the following nephrotoxic medications: nonsteroidal anti-inflammatories, angiotensin-converting enzyme inhibitors, angiotensin II receptor blockers, aminoglycosides, piperacillin/tazobactam, loop diuretics, amphotericin B, acyclovir, intravenous contrast, and nephrotoxic chemotherapy (cisplatin). The category of concomitant nephrotoxic medications was also collected including the continuation of a home nephrotoxic medication vs the initiation of a new nephrotoxic medication.
Statistical Analysis
The primary endpoint of the incidence of vancomycin-induced AKI was compared using a Fisher exact test. The secondary endpoint of the percentage of trough levels or AUC/MICs in the therapeutic, subtherapeutic, and supratherapeutic range were compared using a student t test. The secondary endpoint of first level or AUC/MIC within goal range was compared using a χ2 test. Continuous baseline characteristics were reported as a mean and compared using a student t test. Nominal baseline characteristics were reported as a percentage and compared using the χ2 test. P values < .05 were considered statistically significant.
RESULTS
This study included 97 patients, 43 in the AUC/MIC group and 54 in the trough group.
One (2%) patient in the AUC/MIC group and 2 (4%) patients in the trough group experienced vancomycin-induced AKI (P = .10) (Table 2).