Reducing False-Positive Results With Fourth-Generation HIV Testing at a Veterans Affairs Medical Center
Background: In 2006, the Centers for Disease Control and Prevention issued guidelines advocating routine HIV screening for all patients. However, false-positive results are a potential patient care threat for low-risk populations even with accurate screening assays. A reduction in HIV false-positive screening results can potentially be seen by switching from the third-generation to a more sensitive and specific fourth-generation screening assay.
Methods: We studied the impact on the false-positive screening rate of a change to a fourth-generation assay at a regional US Department of Veteran Affairs Medical Center. HIV screening tests performed by the laboratory from March 1, 2016 to February 28, 2017, prior to implementation of the new assay were compared with fourth-generation HIV screening tests performed from March 1, 2017 to February 28, 2018.
Results: Of 7,516 third-generation HIV screening tests reviewed, 52 were reactive on the screening assay; 24 were true positives, 28 were false positives. The following year 7,802 fourth-generation HIV screening tests were performed and 23 were reactive on the screening assay; 16 were true positives and 7 were false positives. The positive predictive value for the third-generation test was 46% and 70% for the fourth-generation test.
Conclusions: There were fewer false-positive results with testing with the more specific fourth- vs third-generation assay (0.09% vs 0.37%, respectively), which was statistically significant ( P = .002). This reduction in false-positive screening would reduce the laboratory workload and would save an estimated $3,875 yearly and reduce the adverse effects of false-positive screening results for patients.
Results
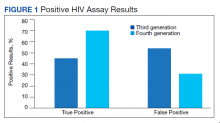
From March 1, 2016 to February 28, 2017, 7,516 specimens were screened for HIV, using the third-generation assay, and 52 specimens tested positive for HIV. On further review of these reactive specimens per the CDC laboratory testing algorithm, 24 tests were true positive and 28 were false positives with a PPV of 46% (24/52) (Figure 1).
From March 1, 2017 to February 28, 2018, 7,802 specimens were screened for HIV using a fourth-generation assay and 23 tested positive for HIV. On further review of these reactive specimens per the CDC laboratory testing algorithm, 16 were true positive and 7 were false positives with a PPV of 70% (16/23).
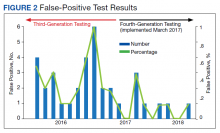
The fourth-generation assay was more specific when compared with the third-generation assay (0.09% vs 0.37%, respectively) with a 75.7% decrease in the false-positivity rate after the implementation of fourth-generation testing. The decreased number of false-positive test results per month with the fourth-generation test implementation was statistically significant (P = .002). The mean (SD) number of false-positive test results for the third-generation assay was 2.3 (1.7) per month, while the fourth-generation assay only had a mean (SD) of 0.58 (0.9) false positives monthly. The decrease in the percentage of false positives per month with the implementation of the fourth-generation assay also was statistically significant (P = .002) (Figure 2).
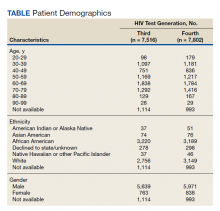
For population-based reference of the tested population at CMJCVAMC, there was a FileMan search for basic demographic data of patients for the HIV specimens screened by the third- or fourth-generation test (Table). For the population tested by the third-generation assay, 1,114 out of the 7,516 total tested population did not have readily available demographic information by the FileMan search as the specimens originated outside of the facility. For 6,402 of 7,516 patients tested by the third-generation assay with demographic information, the age ranged from 25 to 97 years with a mean of 57 years. This population of 6,402 was 88% male (n = 5,639), 50% African American (n = 3,220) and 43% White (n = 2,756). For the population tested by the fourth-generation assay, 993 of 7,802 total tested population did not have readily available demographic information by the FileMan search as the specimens originated outside of the facility. For the 6,809 of 7,802 patients tested by the fourth-generation assay with demographic information, the age ranged from 24 to 97 years with a mean age of 56 years. This population was 88% male (n = 5,971), 47% African American (n = 3,189), and 46% White (n = 3,149).
Discussion
Current practice guidelines from the CDC and the US Preventive Services Task Force recommend universal screening of the population for HIV infection.5,6 As the general population to be screened would normally have a low prevalence of HIV infection, the risk of a false positive on the initial screen is significant.17 Indeed, the CMJCVAMC experience has been that with the third-generation screening assay, the number of false-positive test results outnumbered the number of true-positive test results. Even with the fourth-generation assay, approximately one-third of the results were false positives. These results are similar to those observed in studies involving nonveteran populations in which the implementation of a fourth-generation screening assay led to significantly fewer false-positive results.18