A Robotic Hand Device Safety Study for People With Cervical Spinal Cord Injury
An estimated 282,000 people in the US are living with spinal cord injury (SCI).1 Damage to the cervical spinal cord is the most prevalent. Among cervical spinal cord trauma, injury to levels C4, C5, and C6 have the highest occurrence.1 Damage to these levels has significant implications for functional status. Depending on pathology, patients’ functional status can range from requiring assistance for all activities of daily living (ADL) to potentially living independently.
Improving upper-limb function is vital to achieving independence. About half of people with tetraplegia judge hand and arm function to be the top factor that would improve quality of life (QOL).2 Persons with traumatic cervical SCI may lose the ability to use their hands from motor deficits, sensory dysfunction, proprioception problem, and/or loss of coordination. In addition, they may develop joint contracture, spasticity, pain, and other complications. Thus, their independence and ADL are affected significantly by multiple mechanisms of pathology.
Upper-extremity rehabilitation that emphasizes strengthening and maintaining functional range of motion (ROM) is fundamental to SCI rehabilitation. Rehabilitation to restore partial hand function has included ROM exercises, splinting, surgical procedures in the form of tendon transfers and various electrical stimulation devices, such as implantable neuroprostheses.2-7 These interventions improve the ability to grasp, hold, and release objects in selected individuals; however, they have not been universally accepted. Traditional modalities, such as active ROM (AROM) and passive ROM (PROM) and electrical stimulation remain highly used in upper-extremity rehabilitation. Devices have been developed to provide either PROM or electrical stimulation to improve hand function and to prevent muscle atrophy. Therapist- and caregiver-directed PROM exercises are time consuming and labor intensive. An innovative therapeutic approach that can provide all these modalities more efficiently is needed in SCI rehabilitation.
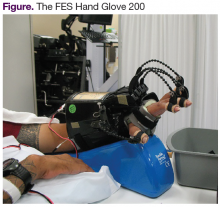
Until now, a single device that combines AROM and PROM simultaneously has not been available. A robotic system, the FES Hand Glove 200 (Robotix Hand Therapy Inc, Colorado Springs, CO), was developed to improve hand function (Figure).
Methods
This prospective safety study evaluated the occurrence of adverse effects (AEs) associated with the use of the FES Hand Glove 200. The study was performed in the Occupational Therapy Section of the Spinal Cord Injury Center at the James A. Haley Veterans’ Hospital (JAHVH) and approved by the JAHVH Research and Development Committee as well as the University of South Florida Investigational Review Board. For recruitment, the goals of the study as well as the inclusion and exclusion criteria were presented to the Spinal Cord Injury Center health care providers. Potential candidates of the study were referred to the study team from these providers.
Screening of the referred candidates was conducted by physicians during inpatient evaluations. All subjects signed a consent form. Participants included active-duty military or veterans with traumatic SCI at levels C4 to C8 and American Spinal Injury Association Impairment Scale (AIS) grades A, B, C, and D. Participants were aged 18 to 60 years, at least 1-month post-SCI, medically stable, and had impairments in upper-extremities strength and ROM or function, including hand.
Subjects were excluded if any of the following were present: seizure within 3 months of study; active cancer; heterotopic ossification below the shoulder; new acute hand injuries of the study limb; unhealed fractures of the study limb; myocardial infarction within 12 months; severe cognitive impairment determined by a Modified Rancho Score below VI8; severe aphasia; pregnancy; skin irritations or open wounds in the study limb; fixed contractures of > 40° of the metacarpophalangeal (MP) or proximal interphalangeal (PIP) joints of the study hand; unwillingness to perform all of the therapies and assessments required for the study; active implant device (eg, pacemaker, implanted cardiac defibrillator, neurostimulator or drug infusion device); major psychological disorder; severe residual spasticity despite maximal medical therapy; muscle power grade of more than 3+ on wrist and finger extensors and flexors of the study limb; recent or current participation in research that could influence study response; pain that prevents participation in the study; or concurrent use of transcutaneous electrical stimulation on the study arm.
The following data were documented: level of SCI, AIS-score; complete medical history; physical examination (including skin integrity); and vital signs of bilateral upper extremities. A nurse practitioner (NP) certified in Functional Independent Measure (FIM) conducted chart reviews and/or in-person interviews of each subject to establish a FIM score before and after 6 weeks of research treatment. Two experienced occupational therapists (OTs) conducted detailed hand evaluations before the research treatment interventions. An OT provided subjects with education on the use, care, and precautions of the FES Hand Glove 200. The OT adjusted the device on the subject’s hand for proper fitting, including initial available PROM, and optimal muscle stimulation.
The OT then implemented the treatment protocol using the FES Hand Glove 200 in 1 hand per the subjects’ preference. The subjects received 30 minutes of PROM only on the FES Hand Glove 200, followed by an additional 30 minutes of PROM with FES for 1 hour of therapy per session. The study participants were treated 4 times per week for 6 weeks. Before and after each session, OTs evaluated and documented any loss of skin integrity and pain. Autonomic dysreflexia occurred when systolic BP increased > 20 to 30 mm Hg with symptoms such as headache, profuse sweating, or blurred vision was reported.9 The FES Hand Glove 200 was set up for PROM to the thumb and to digits 2 to 5 and for electrical muscle stimulation of the finger extensors and flexors. No other therapeutic exercise was performed during the study period on the other extremity. Primary and secondary outcomes were collected at the end of the 6-week intervention.
Primary outcomes included complications from the use of FES Hand Glove 200, including skin integrity and any joint deformity as drawn on a figure, changes of pain level by visual analog scale (VAS), and total number of autonomic dyreflexia episodes. Secondary measured outcomes included changes in PROM and AROM of wrist, metacarpal joint and interphalangeal joints of thumbs and digits 2 to 5 ≥ 10°; hand and pinch strength decline of > 1 lb; decline in manual muscle test, and FIM score, which is a validated measurement of disability and the level of assistance required for ADL.10
Statistical analyses were performed using SAS version 4 (Cary, NC) to assess the degree of change in the improvement score, which was defined as the postintervention score minus the preintervention score. However, because of the large standard error due to small sample sizes, the normality assumption was not satisfied for all the outcomes considered.