Single-Tablet Regimens in the Treatment of HIV-1 Infection
Together with prophylaxis for opportunistic infections, the widespread use of combination antiretroviral therapy (ART)—known as highly active antiretroviral therapy (HAART)—led to substantial gains in the life expectancy of patients infected with human immunodeficiency virus type 1 (HIV-1).1-3 The emergence of HAARTs has decreased mortality, increased viral suppression, and significantly reduced the risk of the sexual transmission of HIV.4-6
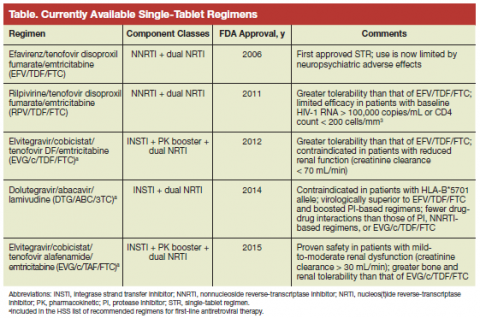
Since its introduction, HAART has evolved significantly with more options for triple therapy combinations that show improved efficacy, convenience, and tolerability. Single-tablet regimens (STRs) are the culmination of this evolution. The first STR was introduced in 2006, and to date, there are 5 STR options approved for use. In this review, the authors discuss the rationale and path to development of STRs, compare the currently available STRs, and discuss practical considerations in their use for the treatment of HIV-1 infection.
Is Triple Therapy The Sweet Spot?
Because current ART cannot eradicate HIV-1 infection due to latently infected CD4+ T-cell reservoirs, the primary goals of ART according to the HHS guidelines are to (1) reduce HIV-associated morbidity and prolong the duration and quality of survival; (2) restore and preserve immunologic function; (3) maximally and durably suppress plasma HIV viral load; and (4) prevent HIV transmission.7
These goals currently seem to be optimally achieved by treatment with 3 active agents, usually consisting of 2 nucleos(t)ide reverse-transcriptase inhibitors (NRTIs) as a backbone plus a drug from 1 of 3 drug classes: an integrase strand transfer inhibitor (INSTI), a nonnucleoside reverse-transcriptase inhibitor (NNRTI), or a protease inhibitor (PI) boosted with ritonavir or cobicistat. The most widely used NRTI backbones are abacavir plus lamivudine (ABC/3TC) or tenofovir disoproxil fumarate plus emtricitabine (TDF/FTC). Attempts to increase the number of active drugs in a regimen (≥ 4) have generally failed to yield additional virologic or immunologic benefits. Conversely, recent attempts to decrease the number of drugs in a regimen (≤ 2) have met with mixed success.
Mega-HAART
The AIDS Clinical Trial Group (ACTG) 5095 was a randomized controlled trial (RCT) to compare the safety and efficacy of a 3-drug regimen (zidovudine/lamivudine [AZT/3TC] plus efavirenz [EFV]) vs a 4-drug regimen (AZT/3TC/ABC plus EFV) in the initial treatment of HIV-1 infection.8 There were no significant differences in virologic efficacy between the 3-drug and 4-drug regimens, findings that have been corroborated in subsequent studies.9,10 Intensified (≥ 4 drugs) regimens have also failed to lead to significant decay in viral reservoir.10,11
Dual Therapy With Newer Agents
Recent clinical studies have explored the use of fewer medications in treatment-naïve HIV-infected patients. In the GARDEL study, dual therapy consisting of lopinavir/ritonavir (LPV/RTV) plus 3TC proved to be virologically noninferior to triple therapy of LPV/RTV plus 2 NRTIs in a fixed-dose combination.12 In a large European clinical trial (NEAT001/ANRS143), the combination of darunavir/ritonavir (DRV/RTV) plus raltegravir (RAL) was noninferior to DRV/RTV plus TDF/FTC, although more virologic failures were seen in the dual-therapy arm for patients with high baseline HIV-1 RNA viral loads or low baseline CD4+ counts.13
Suboptimal virologic responses also were observed with the DRV/RTV plus RAL regimen in several smaller studies: ACTG 5262 and RADAR.14,15 Finally, a combination of DRV/RTV plus maraviroc proved virologically inferior to standard DRV/RTV plus TDF/FTC in the MODERN trial.16 Although some dual-agent regimens have shown promise, none have established consistent and durable virologic efficacy sufficient to supplant triple therapy, which is currently endorsed by treatment guidelines and forms the basis for current STRs.
Current Single-Tablet Regimens
There are currently 5 STRs available for the treatment of HIV-1 infection, 3 are among the recommended regimens for ART in treatment-naïve patients. An older, triple NRTI-based STR (AZT/3TC/ABC) has fallen out of routine use due to inferior virologic efficacy, toxicities, and better-tolerated alternative agents.17 A review of the indications for use, major clinical trials, and unique features of available STRs follows and is summarized in the Table.