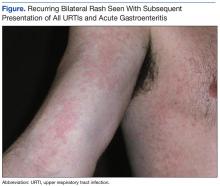
Recurring Bilateral Rash Concomitant With Upper Respiratory Tract Infection in a Healthy Adult Male
A 46-year-old white male presented to a civilian general practitioner with an acute bilateral erythemetous, nonurticarial morbilliform rash. The patient initially presented with these symptoms (7 years prior) to a military field medical clinic, and the first presentation was seen by a nurse practitioner. The rash was evident on the lateral aspects of the thorax and on the medial portions of both arms and upper thighs. There was no evidence of involvement of hands or feet. Outside of being quite prominent, the patient reported the rash to be asymptomatic.
The patient also had concomitant symptoms of a typical upper respiratory tract infection (URTI) with fever of 100.3° F. The physical examination was unremarkable except for the pronounced rash as described and URTI-associated nasal congestion/postnasal drainage. Examination of oral mucosa of the head, ears, eyes, nose, and throat also was negative for any evidence of eruption. The patient was in excellent physical condition, took no routine medications, and had no documented underlying medical conditions. His family and social history seemed noncontributory; he reported frequent domestic airline travel associated with reserve duty as well as with his civilian job. Appropriate immunizations were all current, and regularly scheduled physical examinations had been documented in the medical record.
The patient’s rashes associated with URTI began 7 years prior at age 39 after a weeklong (fall season) operational readiness inspection (ORI), which included extended periods wearing ground crew ensemble (GCE) equipment. Protective clothing worn during that ORI included the heavily charcoal-laden over-garment, butyl rubber gloves and boots, and the then current-issue gas mask. The rashes had never appeared before age 39, and the patient reported no previous medical problems after wearing GCE equipment during 16 years of service preceding that initial presentation.
The patient’s rashes typically presented with the similar pattern and intensity at 24 to 36 hours after onset of URTI symptoms, were self-limiting, and resolved in an additional 48 to 72 hours (Figure). The rash was present with each subsequent URTI and occurred on average, about 2 times each year. Similar rashes also appeared twice during the preceding 7 years during episodes of nausea/vomiting/diarrhea consistent with acute gastroenteritis (AGE). No other household members (infant to aged 40 years) showed any similar rash during episodic URTI or AGE during the same period. However, about 2 years after the initial URTI/rash presentation, the patient’s then toddler-aged son was diagnosed with self-limiting unilateral laterothoracic exanthem (ULTE), which completely resolved in 2 to 3 weeks and never reappeared.
Discussion
The author postulated that the patient most likely experienced routine acute episodes of Picornaviridae infection. The overarching diagnosis was presumptive, as biopsy and/or serologies were not obtained on the reservist. The Picornavirus family includes genera such as Rhinovirus and Enterovirus, both significant contributors to URTI (Enterovirus for AGE). Typically, about 50% of URTIs are caused by Rhinovirus (approaches 100% in some seasonal outbreaks) with the remainder of common URTIs being caused by Enterovirus, Coronavirus, Adenovirus, Reovirus, and Parainfluenza virus.1 Unfortunately, rapid office-based or field medicine diagnostic assays to consistently confirm or differentiate these common URTI viral etiologies (excepting influenza A/B) are simply not economically feasible at this time nor are any standardized laboratory tests recommended for self-limiting URTIs in immunocompetent adults. There are multiplex polymerase chain reaction assays available for many viral URTI pathogens, but cost and the need for laboratory as well as certified medical technologist support may often preclude routine use at the point of care.
Perhaps the more intriguing aspect of this case is the consistent recurrence of the same rash presentation in a middle-aged immunocompetent adult male with every subsequent episode of typical URTI or AGE over a period of 7 years, irrespective of seasonal timing, contact history, or geographic location. There is well-documented evidence of URTI associated with Adenovirus and Coronavirus seen in otherwise healthy adults living in close quarters military environments. Particularly, Adenovirus serotypes 3, 4, 7, and 21 have been associated with respiratory disease in military troop populations.2
There are also numerous viral exanthems shown in young children with probable links to Epstein-Barr virus (EBV), Human herpesvirus (HHV) 6, and parvovirus B19.3 The patient’s then toddler-aged son experiencing ULTE subsequent to the father’s presentation raise the question of a possible link to viruses other than those associated with typical URTI/AGE. Literature review indicated ULTE may be linked to EBV, parvovirus B19, Adenovirus, Parainfluenza virus, or HHV 6 and 7.4
Adenovirus and Parainfluenza virus are causative agents for a portion of URTIs as well. However, the likelihood of all of the patient’s subsequent URTIs being caused exclusively by Adenovirus would be exceedingly low. Infections with atypical bacteria, such as mycoplasma, also are frequently associated with rash, fever, and myalgia but are more commonly linked to lower RTI.
All the above etiologies are typically self-limiting (except mycoplasma infections) and usually resolve without specific treatment. This broad differential tends to “muddy the water” when trying to tie a definitive diagnosis to what was a seemingly routine URTI in a healthy adult.
The patient’s status as a member of the reserve complicated obtaining a definitive diagnosis of the recurring rash. Even if a link between the recurring rash and earlier duty could be established, access to an acute-care appointment at a military treatment facility (in nonduty status) was/is not authorized. A timely civilian acute-care appointment for routine URTI with short-lived rash in an adult patient is difficult to book, especially if the rash appears on a weekend or holiday. Likewise, insurance reimbursement for skin biopsy or numerous serologic tests (investigating possible viral etiology) is frequently not covered for such conditions, as even a positive serology likely would not alter the current standard of supportive care.
Evaluation of any patient presenting during or immediately after field exercises or operational deployment must be conducted in full consideration of such conditions. A thorough history relating to geographic location, immunization status, season, environmental or chemical contacts, animal/insect exposure, living conditions, other ill unit personnel, sanitation/hygiene and food/water quality is of paramount importance. Likewise, there may be value in at least having a unit briefing/follow-up with a medical provider 2 to 6 months after deployment/field training activities. A general follow-up such as this may help find any emerging trends or common concerns pertaining to unit members’ health.
Reservists present a unique health maintenance challenge as they may complete a duty tour and drop out of military health system visibility for weeks to months. The inherent design of some reserve component programs makes even the discovery of a case such as this difficult at best. A structured follow-up (be that a group briefing or even a survey-type questionnaire) becomes highly useful for monitoring emerging health trends in reservists.