UPDATE ON CERVICAL DISEASE
Will co-testing further reduce your patients’ risk of cervical Ca, compared with current Pap screening alone? How should you care for a woman who has a Pap–/HPV+ result? Here are answers, illuminated by new data.
IN THIS ARTICLE
FIGURE 1 Cervical adenocarcinoma in situ
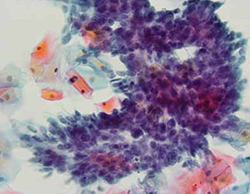
Cervical AIS demonstrated in a cytology specimen. Most studies have shown that cytology is less sensitive than HPV testing for a glandular lesion.
Photo: Courtesy of Dennis O’Conner, MD.
Similar results were described by Katki and coworkers in a large HMO screening population in the United States, in which Pap–/HPV+ women had half the risk of cancer over the subsequent 5 years, compared with women who were negative by Pap testing only (3.2 cases for every 100,000 women, compared with 7.5 cases, respectively). Of the total cases of CIN 3+ found, 35% of cases of CIN 3 and adenocarcinoma in situ (AIS) (FIGURE 1) were detected in women Pap-/HPV+, as were 29% of cancers. Cytology has been shown, in almost all studies, to be less sensitive for glandular lesions; it isn’t surprising, therefore, that 63% of adenocarcinomas found in this study population were detected by HPV testing only.
These results appear to provide overwhelming evidence of the benefit of including HPV testing in screening programs for cervical cancer.3
Adding HPV testing to primary screening with Pap testing (co-testing) appears to reduce the long-term risk of cervical Ca. Consider making this stratagem part of your practice.
Dilemma: How do we best manage women who are Pap–/HPV+?
Kinney W, Fetterman B, Cox JT, Lorey T, Flanagan T, Castle PE. Characteristics of 44 cervical cancers diagnosed following Pap-negative, high risk HPV-positive screening in routine clinical practice. Gynecol Oncol. 2011;121(2):309–313.
Although co-testing appears to reduce the risk of cervical cancer—allowing a safer margin for Pap-negative plus HPV-negative (Pap–/HPV–) women if they miss by up to 2 years their 3-year recommended screening—the question remains: What is the best way to manage Pap–/HPV+ women?
The primary recommendation by both the American Society for Colposcopy and Cervical Pathology (ASCCP) and ACOG is to re-screen them in 12 months, rather than send them immediately for colposcopy. Why? Because HPV infection 1) is relatively common among women who do not have CIN 2, 3 or invasive cervical cancer and 2) most often resolves without causing significant disease.
But re-screening these patients in 12 months negates much of the benefit of the high clinical sensitivity of HPV testing because it could lead to significant delay in the diagnosis and treatment of some women who already have either cervical cancer or advanced CIN 3 that might progress to cancer before their next evaluation. This concern is supported by the findings of the studies I discussed earlier.1-3
Although cervical cancer missed at cytology screening is uncommon, 44 cervical cancers were reported by Kinney and coworkers in a large screening population in women who had one or more Pap–/HPV+ co-test results (18 had two or more Pap–/HPV+ results before diagnosis). More than 60% had cervical adenocarcinoma, which is more than three times the normal proportion of glandular cancer to squamous cervical cancer—again highlighting the relative insensitivity of cytology to detect glandular lesions.
Other drawbacks of waiting 12 months for further evaluation include 1) prolonged uncertainty for the patient and 2) the potential for losing her to follow-up, which occurs in as many as 50% of subjects in many studies.
Therefore, finding patients who are at greatest risk of CIN 2+ among the larger group of Pap–/HPV+ women, and referring them immediately for colposcopy, should provide great benefit.
Note that the 2006 ASCCP guidelines included a statement that, once an FDA-approved test for HPV 16, 18 was available, triage of Pap–/HPV+ patients who are positive for HPV 16, 18 could identify a majority at greatest risk of CIN 2,3+ and who would therefore benefit most by referral for colposcopy. In contrast, patients at lower risk (i.e., positive for any of the other 12 HPV types but not for types 16 and 18) would be better managed by repeating co-testing in 12 months—referring for colposcopy only those who are found again to be HPV+ or who have an abnormal Pap test.
In the next section of this Update, I explore recent evidence about the important role played by HPV 16, 18 in cervical carcinogenesis.
ASCCP guidelines provide the option of testing Pap–/HPV+ women for HPV 16, 18 and referring women positive for either of these types to colposcopy.
HPV 16, 18 pose long-term risk for CIN 3+ and cervical Ca
Kjaer SK, Frederiksen K, Munk C, Iftner T. Long-term absolute risk of cervical intraepithelial neoplasia grade 3 or worse following human papillomavirus infection: role of persistence. J Natl Cancer Inst. 2010;102(19):1478–1488.







