INFECTIOUS DISEASE
New data shed light on preventing cytomegalovirus infection and wound complications, and reveal a disturbing picture of Clostridium-related diarrhea in pregnant women.
IN THIS ARTICLE
Patients who receive a broad-spectrum antibiotic should be monitored closely for signs of toxicity; if diarrhea develops, discontinue the offending drug
Dr. Duff reports no financial relationships relevant to this article.
Four important developments have marked the past year in infectious disease:
- A promising vaccine against cytomegalovirus (CMV) was tested in women of reproductive age
- Extended-spectrum antibiotic prophylaxis proved to be effective in reducing the incidence of wound infection following cesarean delivery
- Investigators developed a simple but effective method to prevent wound complications following repair of a third- or fourth-degree perineal laceration
- The incidence of severe Clostridium difficile-associated diarrhea crept upward, emerging as a threat to pregnant women.
CMV vaccine makes an auspicious debut—but isn’t ready for practice
Pass RF, Zhang C, Evans A, et al. Vaccine prevention of maternal cytomegalovirus infection. N Engl J Med. 2009; 360:1191–1199 [Classification of evidence – Level I].
This Phase-2, randomized, double-blind, placebo-controlled trial of a new cytomegalovirus (CMV) vaccine in women found an overall efficacy rate of 50% (95% confidence interval, 7% to 73%), with no unusually serious reactions among women who were vaccinated. This efficacy rate is disappointing, but it isn’t entirely surprising; even the immune response resulting from natural infection is not fully protective against reactivated infection or recurrent infection with a different strain of virus. Nor is natural immunity completely effective in preventing severe fetal injury in recurrent infection.1
Virus poses greatest risk to pregnant women and their fetuses
CMV is the most common perinatally transmitted infection, affecting 0.6% to 0.7% of infants ( FIGURE 1 ). The greatest risk of fetal injury occurs when the mother develops primary infection during pregnancy, which raises her infant’s risk of infection to 40% to 50%. Of infants delivered to mothers with primary infection, approximately 10% to 15% will be acutely symptomatic at birth.
Clinical manifestations of severe congenital CMV infection include growth restriction, microcephaly, ventriculomegaly, intracranial calcification, chorioretinitis, hearing impairment, hepatitis, and thrombocytopenia.
Because the morbidity and mortality associated with these conditions are alarmingly high, development of a safe, effective vaccine against CMV would be most welcome.2,3
Recurrent or reactivated maternal CMV infection poses a much lower risk to the fetus. Infected infants are rarely symptomatic at birth. Clinical manifestations of infection typically occur later in childhood and include hearing and visual deficits, dental anomalies, and learning or behavioral disorders.2,3
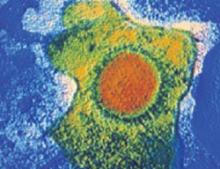
FIGURE 1 Cytomegalovirus
Cytomegalovirus is a member of the herpesvirus family. It is shed intermittently in bodily fluids, without detectable signs and symptoms.
Details of the trial
Women were eligible for the study if they were seronegative for CMV antibody, in good health, 14 to 40 years old, and not pregnant or lactating. Participants received three doses of vaccine or placebo at 0, 1, and 6 months. (The vaccine was composed of CMV envelope glycoprotein B with MF59 adjuvant.) The women were then tested for CMV infection every 3 months for as long as 42 months, using an assay for IgG antibodies directed against viral proteins other than glycoprotein B. Infection was confirmed by viral culture or immuno blotting. The primary endpoint was time until detection of CMV infection.
The vaccine was given to 234 patients, and 230 received placebo. Eighteen infections occurred in the vaccine group, compared with 31 in the placebo group. Vaccinated patients were more likely to remain uninfected during follow-up (p=.02).
One of 81 infants (1%) born to mothers in the vaccinated group had congenital CMV infection, compared with three of 97 (3%) infants born to mothers in the placebo group (p=.41). One infant in the placebo group had severe infection that was evident at birth. The other three infants were asymptomatic at birth and free of sequelae 3 to 5 years later.
The most promising preventive remains experimental
No drug is uniformly effective in treating maternal CMV infection and preventing congenital infection. The most promising intervention for prevention of congenital CMV infection is administration of hyperimmune anti-CMV antibody to the mother. A recent report by Nigro and colleagues4 found this agent to be of great value for both treating and preventing congenital CMV. However, because of limitations in the design of this study, administration of hyperimmune globulin still must be regarded as experimental.3
Key questions remain unanswered
The goal of a large-scale vaccination program is to ensure that women enter reproductive age with preexisting immunity to infection. In that light, the study by Pass and colleagues is only partially encouraging. Despite vaccination, 18 infections occurred, and the follow-up period was relatively short. We do not yet know whether the protective effect of the vaccine will be of extended duration. Moreover, one vaccinated mother delivered an infant who had congenital CMV infection.