Helping patients kick the "other" habit
Chew users want medications—not just counseling. What’s worth trying when the evidence is limited?
TABLE 3
Usefulness of bupropion for smokeless tobacco cessation is unclear
| STUDY (YEAR) | POPULATION | INTERVENTION | COUNSELING | DURATION | RESULTS | NNT |
|---|---|---|---|---|---|---|
| Berigan12 (1999) | 1 can/day | • Bupropion | • Behavioral counseling | 10 weeks | Patient in case report remained ST free at 8 months | N/A |
| Glover13 (2002) | 70 patients • ≥18 year-old- men • ≥½ can/day | • Bupropion SR • Placebo | • Telephone sessions (weeks 9-11) | 3 months | Patients receiving active NRT had significantly higher quit rates by week 7 (P=0.04) | 4.3 |
| Dale14 (2002) | 68 patients • ≥18 years • Regularly used ST | • Bupropion SR • Placebo | • 10 minute behavioral counseling | 24 weeks | No significant difference | N/A |
| NNT, number needed to treat; NRT, nicotine replacement therapy; SR, sustained release; ST, smokeless tobacco. | ||||||
The second trial14 evaluating the efficacy of bupropion for smokeless tobacco cessation was also a randomized, placebo-controlled trial. Enrollees were randomly assigned to bupropion SR 300 mg/day or matching placebo for 12 weeks. In addition, minimal behavioral intervention was provided until week 24. Researchers enrolled 68 patients; all but one were men. Participants in this study were 18 years of age or older who regularly used smokeless tobacco. The efficacy of bupropion SR for nicotine dependence was the primary endpoint evaluated.
At the completion of the treatment phase, more patients in the bupropion group reported cessation (44%) compared to placebo (26%). The difference between the 2 groups was nonsignificant and remained so until the end of the study. By study end, each group had a reported cessation rate of 29%.
One patient experienced a diffuse skin rash that resulted in discontinuation of the study medication; otherwise buproprion was well tolerated.
A product that shows promise for smoking cessation is varenicline, which was approved by the FDA in May 2006.15 Varenicline acts as an agonist at the α4β2 neuronal nicotinic acetylcholine receptors. Its action at this receptor subtype blocks the ability of nicotine to bind to the receptor and is therefore thought to blunt the feeling of reward experienced by smokers.16 In addition, the medication may possess some nicotine-like action at the nicotinic receptor that may decrease the amount of withdrawal symptoms experienced by patients.15
In theory, this mechanism of action may help smokeless tobacco users to succeed in their quit attempts. Unfortunately, though, there have been no published case reports or clinical trials to date regarding the use of varenicline in the treatment of smokeless tobacco cessation.
Helping patients despite the information gap
Our knowledge of the effectiveness of various tools in smokeless tobacco cessation is hampered on a number of fronts.
Nicotine replacement therapy. The conflicting evidence regarding nicotine replacement therapy may be the result of a number of factors. First, although many of the trials used a randomized, placebo-controlled design, there were a number of variations in the patient populations studied, inconsistencies in the amount and type of counseling used, and differences in the amount of smokeless tobacco used by the patients.
These 2 cases illustrate the damage that chew can do
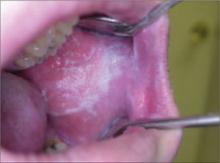
Oral leukoplakia
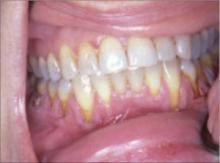
Gum recession
This 24-year-old patient sought treatment for an asymptomatic oral lesion that he noticed while brushing his teeth. The patient, who did not smoke tobacco and drank alcohol socially on weekends, told us that he had started chewing tobacco while playing baseball in high school 8 years earlier. He said that over the past 3 years, he had increased the habit.
On examination, we noted a well-defined, macular, white lesion on his left buccal mucosa. The lesion was not painful with palpation and remained unchanged when scraped with a tongue depressor.
Our patient had an oral leukoplakia, which can result from the chronic use of chewing tobacco. Cessation of the habit typically results in the resolution of the lesion in approximately 4 weeks. That was the case with our patient: He quit the habit and in 4 weeks, 80% of the lesion resolved. He was closely monitored, and at 3 months the lesion was undetectable.
If, however, a patient discontinues the habit and there is no change in the lesion after 4 weeks, biopsy is indicated. The most serious consequence of a malignant transformation of leukoplakia is oral squamous cell carcinoma.
A 30-year-old man was referred to us for evaluation of gum recession that had worsened over the past year. The patient complained that his teeth were sensitive to hot and cold drinks, but had no other symptoms. He had a 5-year history of smokeless tobacco use and said he usually placed the tobacco along his inner vestibule. He said that he did not smoke tobacco, nor did he drink alcohol.
On examination, we noted that the patient had localized recession along the cervical area of his lower teeth. With manipulation, there was bleeding from the gingival surface. His teeth were otherwise in good shape and there were no other lesions within the oral mucosa.
Gingival recession is a common consequence of smokeless tobacco use. Treatment consists of a gingival graft, in which palatal connective tissue is removed and used to reestablish gingival attachment to the tooth. (Tissue harvesting surgery from the palate can be quite painful.) Left untreated, teeth can become loose and fall out.
We advised our patient to follow up for gingival grafting, but he did not return for his follow-up visit.
Denise Rizzolo, PA-C, PhD is a physician assistant at Care Station in Springfield, NJ and Thomas A. Chiodo, DDS, is an oral and maxillofacial surgeon in private practice in Somerville, NJ. Dr. Chiodo also teaches at UMDNJ Dental School in Newark, NJ; rizzoloden@aol.com