Diagnosis and 10-Year Follow-Up Of a Community-Based Hepatitis C Cohort
During the period of observation, which ended December 31, 1999, 49 subjects (14%) received interferon treatment specifically for their hepatitis C. Half of subjects (n = 25) received that treatment during clinical trials. Twenty people (12 in clinical trials) received ribavirin in addition to interferon; 1 received interferon and interleukin.
Many of the other 306 subjects had 1 or more documented contraindications to HCV therapy (Table 2). The 53 instances of chemical dependency may underestimate the effect of chronic alcohol ingestion on decisions not to treat, since among the 355 subjects, documentation of chronic heavy alcohol consumption (>6 drinks/day) was listed in the medical records of 182 (51%). The total number of subjects with one or more conditions that might be considered contraindications to therapy was 225 (63%). Although only a few of these contraindications were permanent conditions (eg, vegetative state) (Table 2), no subjects had a repeat reference to treatment after the documented condition (eg, depression) had resolved or improved.
Information on potentially accelerating comorbid conditions was available in many charts (ie, those of 11 subjects with known HIV infection). No HIV testing was documented, however, in 55 people who had HCV risk factors other than blood transfusion. Five people had documented chronic HBV infections (surface antigen positive). HBV screening was almost universal. Yet 159 subjects were not immune (including no documented HBV immunizations), and of these, 108 had HCV risk factors other than transfusion before 1992 and were therefore eligible for HBV immunization. Hepatitis A vaccination is now recommended for all nonimmune HCV patients,5 but immunization for hepatitis A was documented for only 25 subjects.
TABLE 1
RISK FACTORS NOTED AT DIAGNOSTIC VISIT
| Risk Factor | No. of Patients (%) (N=355) |
|---|---|
| History of intravenous drug use | 177 (50%) |
| Sexual exposure | 128 (36%) |
| Immigrant | 47 (13%) |
| Tattoos | 60 (17%) |
| History of blood transfusion | 107 (30%) |
| Occupational risks | 44 (12%) |
TABLE 2
REASONS DOCUMENTED FOR NOT RECEIVING TREATMENT
| Reason | No. of Patients (%) (N=306) |
|---|---|
| No reason stated | 81 (26%) |
| Chemical dependency | 53 (17%) |
| Comorbid condition | 28 (9%) |
| Refused/noncompliant | 44 (14%) |
| Age | 13 (4%) |
| Ineligible | 22 (7%)* |
| Depression | 25 (8%) |
| Psychiatric condition | 4 (1%) |
| Desired pregnancy | 5 (2%) |
| Cirrhosis | 6 (2%) |
| Hepatocellular carcinoma | 8 (3%) |
| HIV treatment | 7 (2%) |
| No insurance | 6 (1%) |
| No trial available | 4 (1%) |
| *Ineligible because of advanced liver disease or other terminal illness. | |
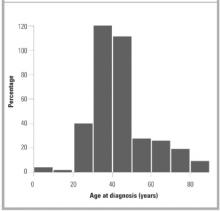
FIGURE 1
AGE AT DIAGNOSIS
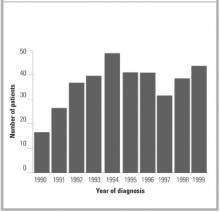
FIGURE 2
NUMBER OF NEW DIAGNOSES PER YEAR
Discussion
Hepatitis C was an uncommon new diagnosis in Olmsted County and therefore not a frequent occurrence in the practice of any of the 200 primary care physicians in the community. Although the overall prevalence of hepatitis C was only about 25% of that reported for the Midwestern United States (1.3%),3,4 the ratio of men and women diagnosed with HCV and the distribution of recorded risk factors in our cohort are similar to those reported from the NHANES conducted in 1988 to 19944 and to those in other population-based studies.5,15-17
In this cohort, primary care physicians’ response to known HCV varied from occasional monitoring of AST/ALT tests to referral for specialty evaluation and HCV treatment. In a significant group of patients, no visits for HCV follow-up could be identified. The only other published data on primary care physicians’ follow-up care of patients with known HCV was self-report survey data.18 In response to a survey, primary care physicians reported they ordered yearly AST/ALT tests in all HCV patients and referred over 80% of people with known HCV to hepatologists. Self-reported care often overestimates the amount of care provided. This difference in reported and observed care emphasizes the importance of data on actual practice.
The community physicians’ lack of a uniform or aggressive approach to HCV infections may not be surprising in view of the wide disparity of available information on chronic HCV infection and its progression to symptomatic or progressive liver dis-ease.18 For example, published rates of progression to “chronic infection” after exposure vary from 85% of people receiving HCV-infected blood transfusions to 20% of women given HCV-contaminated RhoGAM.9 Even rates of progression in people with persistent viremia (+PCR) vary from 69 % to 88%.19 Unfortunately, progression to chronic HCV infection cannot be predicted from initial clinical or laboratory factors.20 In addition, the meaning of “chronic infection” is unclear.19 Reported rates of progression from chronic infection to cirrhosis are widely divergent and appear related to the type of group used to make predictions. Data from meta-analyses of clinical trial patients21 suggest progression rates of up to 69% in 30 years. Yet only 2.4% to 6% of community-based hepatitis C patients may develop cirrhosis after 17 to 40 years of follow-up.22,23
Wide variations in the progression of cirrhosis to decompensated cirrhosis have been reported.24 Studies of AST levels in people with known HCV show fluctuations over time, dispelling the idea that once elevated, AST or ALT will remain elevated or that AST levels are directly predictive of progression or resolution of liver injury or viral loads.25,26 Chronically elevated AST/ALT levels, however, have been shown to predict progression,27 making monitoring of AST/ALT important in all subjects.5,28