Probiotics as a Tx resource in primary care
While probiotics have not been marketed as drugs, clinicians can still recommend them in an evidence-based manner.
PRACTICE RECOMMENDATIONS
› Consider specific probiotics to prevent antibioticassociated diarrhea, reduce crying time in colicky infants, and improve therapeutic effectiveness of antibiotics for bacterial vaginosis. A
› Consider specific probiotics to reduce the risk for Clostridioides (formerly Clostridium) difficile infections, to treat acute pediatric diarrhea, and to manage symptoms of constipation. B
› Check a product’s label to ensure that it includes the probiotic’s genus, species, and strains; the dose delivered in colony-forming units through the end of shelf life; and expected benefits C
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
We are in the age of the microbiome. Both lay and scientific press proliferate messages about the importance of the microbiome to our health even while they often remain unclear on how to correct microbiota patterns associated with different diseases or suboptimal health states. Probiotics are defined as “live microorganisms that, when administered in adequate amounts, confer a health benefit on the host.”1
Certain probiotics have been shown to prevent and treat specific diseases or conditions, inside or outside the gut. But the level and quality of evidence varies greatly. In addition, the health claims allowed by government regulators depend on making discrete distinctions (food vs drug, maintaining health vs treating disease, and emerging evidence vs significant scientific agreement) along dimensions that are increasingly recognized as continuous and complex.2 This leads to confusion among doctors and patients about whether to trust claims on product labels and what to make of the absence of such claims.
Find out which probiotic is effective for a patient’s condition. Simply recommending that a patient “take probiotics” is not particularly helpful when the individual wants a product that will aid a specific condition. While probiotics, to date, have not been marketed as drugs in the United States, clinicians can still approach recommending them in an evidence-based manner.
In this article, we review diseases/conditions for which probiotic products have good efficacy data. We discuss probiotic efficacy and safety, offer relevant information on regulatory categories of probiotics, and give direction for proper usage based on the current evidence base. Although this review is meant to be an easy-to-use resource for clinicians, it is not a comprehensive or detailed review of the numerous probiotic products and studies currently available.
Regulatory and commercial variances with probiotics
In the United States, probiotics have been marketed as dietary supplements, medical foods, or conventional foods, all of which require different levels of evidence and types of oversight than drugs. The efficacy of some probiotics in treating or preventing certain diseases and conditions is similar to, if not better than, effects observed with traditional drug interventions (TABLE 13-32). However, unlike drugs, which are subject to premarket oversight, the probiotic marketplace contains products with uneven levels of evidence, from well substantiated to greatly limited. Currently, no probiotics are sold in the United States as over-the-counter or prescription drugs, although probiotic drugs will likely enter the US market eventually.
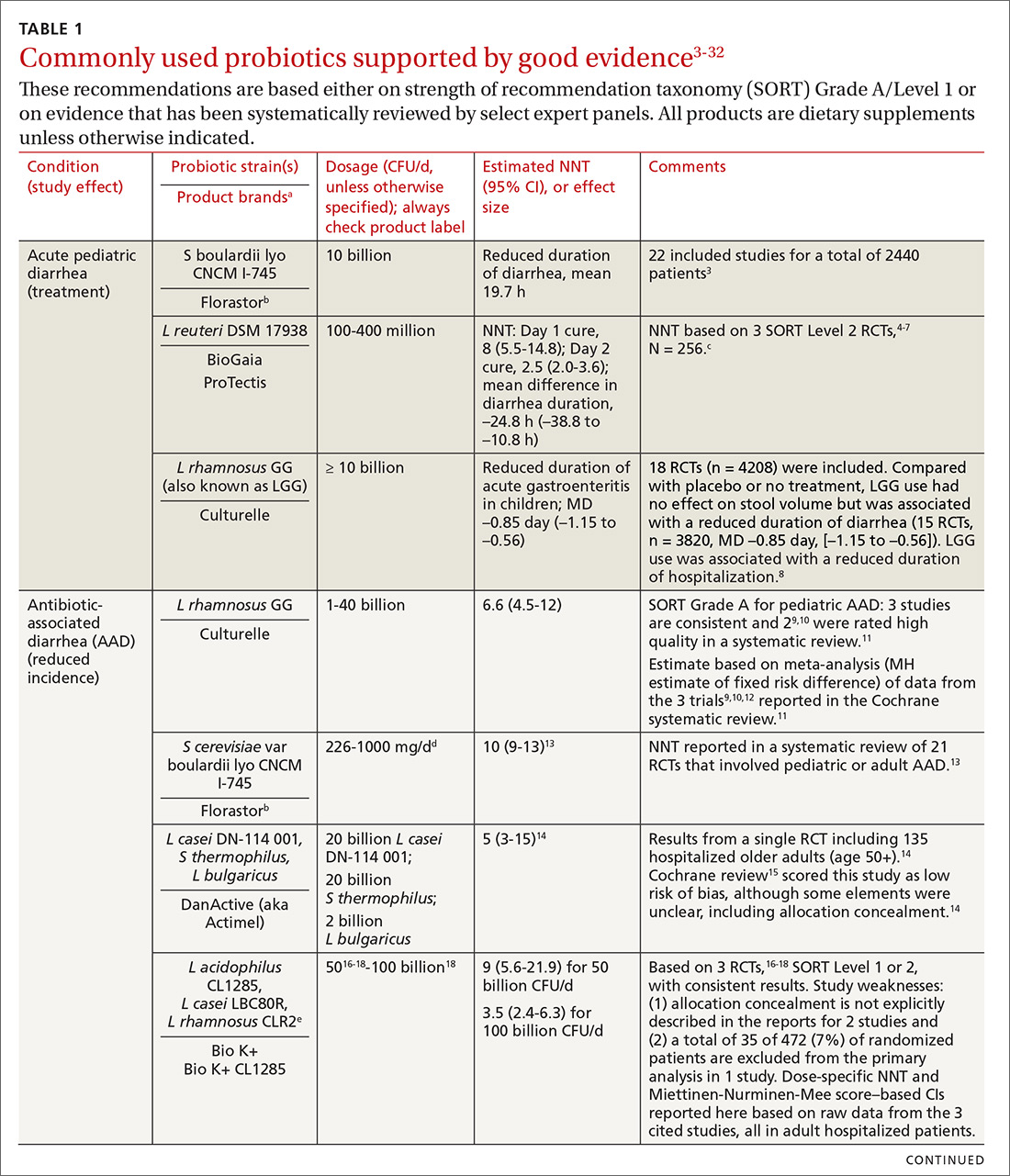
What to consider when recommending a product. When considering probiotics, remember that strain, dosage, and indication are all important. Just as we know that not all antibiotics are equally effective for all infections, so, too, effectiveness among probiotics can—and often does—vary for any given condition. Effectiveness also may vary from patient to patient. Most recommendations made in this review are tied to specific probiotic strains and doses. In some cases, more than one probiotic may be efficacious, likely due to the same or similar underlying mechanism of action. For example, most probiotics produce short-chain fatty acids in the colon, providing a common mechanism supporting digestive health.33-35
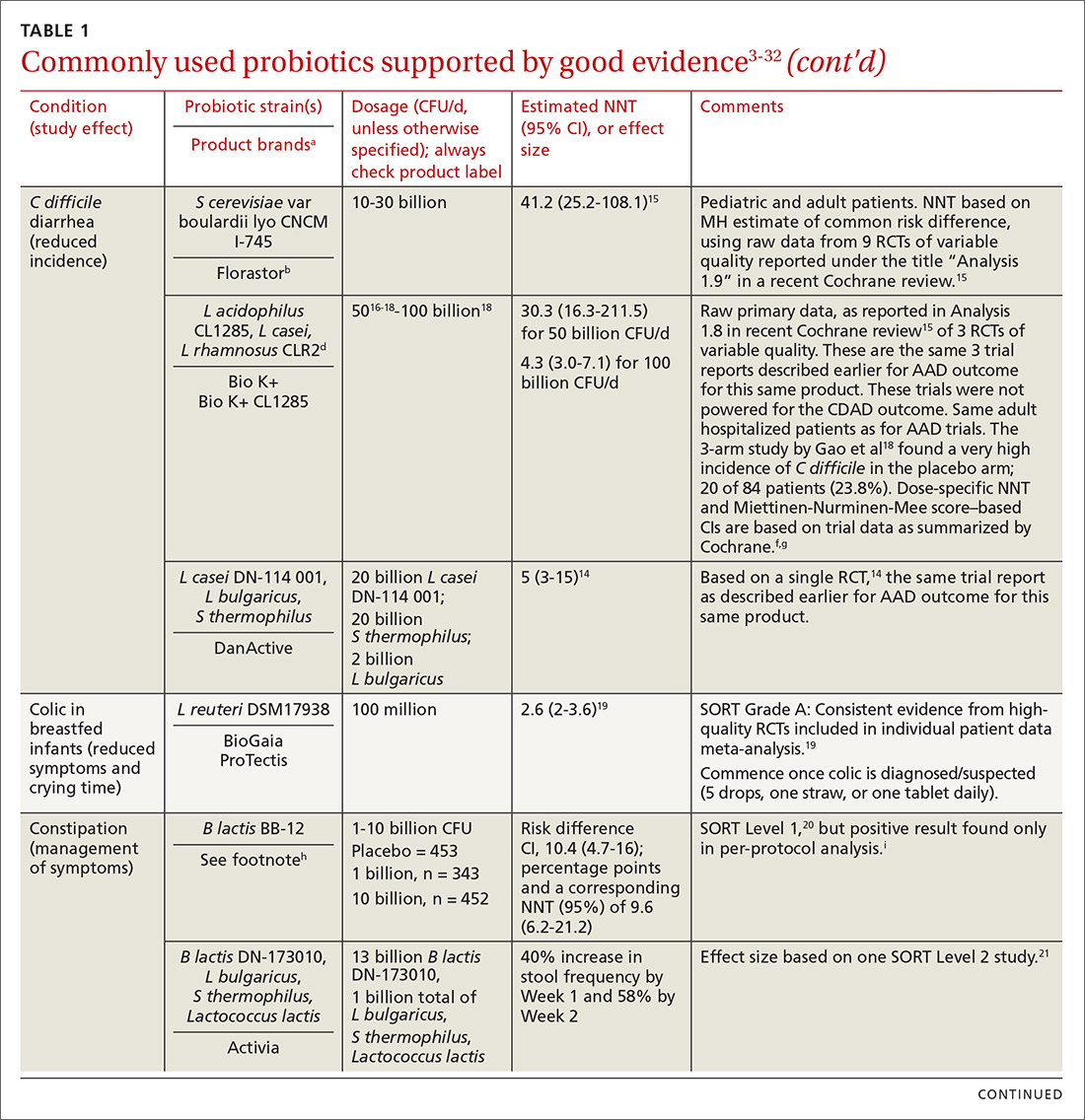
Contrary to the blanket recommendation preferring higher dosages or a greater number of strains,36 our recommendations are based on levels shown to be effective in clinical trials, which in some contexts can be as low as 100 million colony-forming units (CFU) per day.37,38 Indeed, a survey we conducted previously of retail dietary supplement products indicated that products with lower CFUs or fewer strains could more readily be linked to evidence of efficacy than multistrain, high-CFU products.39
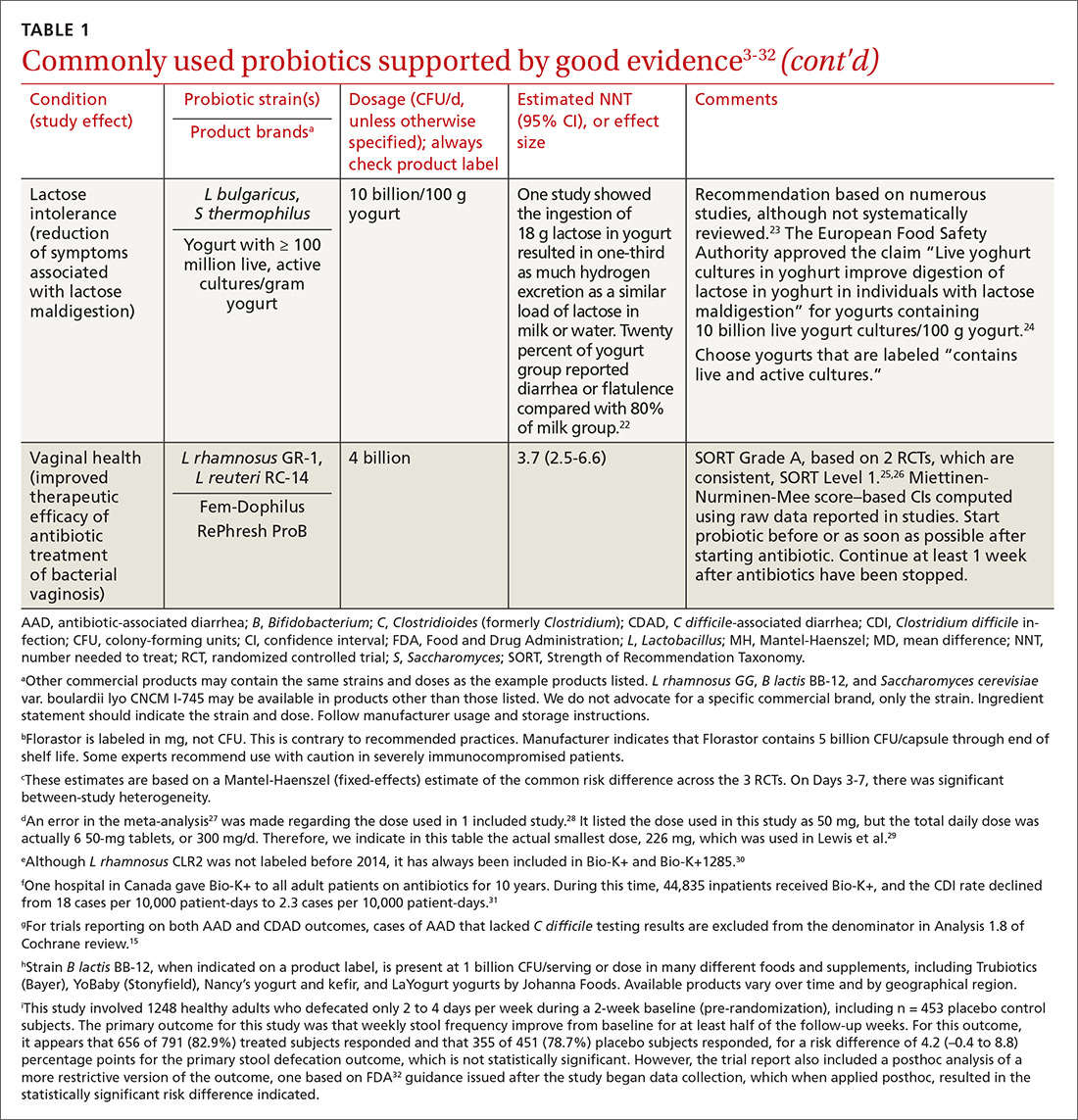
Continue to: Understanding probiotic product labels is a good start






