DECAAF points way to improved AF ablation
AT HEART RHYTHM 2013
DENVER – Preprocedural quantification of atrial fibrosis via delayed-enhancement MRI offers a potent means of boosting atrial fibrillation ablation success rates by weeding out likely nonresponders, a study has shown.
Results of the double-blind, multinational prospective DECAAF trial (Delayed-Enhancement MRI Determinant of Successful Catheter Ablation for Atrial Fibrillation) show that the extent of atrial fibrosis is a strong predictor of patient outcome. The more fibrosis present when measured preprocedurally by delayed-enhancement MRI (DE-MRI) with gadolinium contrast, the less likely a patient would remain in sinus rhythm at follow-up more than 1 year following ablation, Dr. Nassir Marrouche reported at the annual meeting of the Heart Rhythm Society.
"Hopefully this will help us better individualize the therapy. We have to accept at some point that we can’t cure everybody with atrial fibrillation. This modality helps us to better define which patients should be eligible," according to Dr. Marrouche, director of the comprehensive arrhythmia research and management center at the University of Utah, Salt Lake City.
One attractive feature of this management strategy is that patients who are candidates for AF ablation already routinely undergo a preprocedural 3-D scan with CT or MRI. It’s a straightforward matter to train an MRI technician to measure atrial fibrosis, as DECAAF showed.
This is a practice-changing development, he said: "We recommend that in every single ablation patient, before you touch their heart you really should know how extensive their disease is."
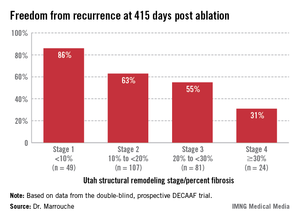
Patients undergoing a first ablation procedure for paroxysmal AF typically have about a 60% success rate, meaning they remain free of atrial arrhythmia at 1 year. In contrast, DECAAF participants with less than 10% fibrosis in their left atrium had a success rate of 86% at 415 days of follow-up.
Patients with less than 10% preprocedural atrial fibrosis are categorized as Utah stage 1 disease using a left atrial structural remodeling classification system previously described by Dr. Marrouche and his coworkers . At the other end of the spectrum, DECAAF subjects who were Utah stage 4, with at least 30% fibrosis, had a dismal 31% freedom of recurrence 415 days after the end of the standard 90-day postprocedural blanking period. Patients with stage 2 or 3 atrial fibrosis had intermediate long-term outcomes (see box).
"We sit down with a stage 4 patient and say, ‘Listen, your chance of being cured of this disease at long-term follow-up is not very high,’" the electrophysiologist explained. "We put our expectations on the table. We now have 5-year follow-up data showing a 5%-10% success in Utah 4 patients. We in Utah today are very reluctant to take such patients into the lab and ablate them. Instead, we try every option to rate-control them. But Utah stage 1 is straightforward. I make that decision for the patient: ‘You should be ablated early on.’ "
DECAAF included 261 patients with AF who underwent a first ablation procedure at 15 centers in the United States, Europe, and Australia. But first they all underwent DE-MRI, the results of which were sent to a core lab for blinded quantification of atrial fibrosis. The interventionalists remained blinded to the fibrosis findings.
Most DECAAF participants had paroxysmal AF. The ablation procedure consisted of pulmonary vein isolation alone in 68% of patients and pulmonary vein isolation plus other ablation maneuvers in the rest.
The only predictor of atrial fibrosis on DE-MRI was a history of hypertension. Duration of AF, patient age, AF type, and comorbid conditions were not associated with the extent of fibrosis.
In a multivariate analysis controlled for numerous potential confounders, the only significant predictor of AF recurrence was atrial fibrosis. For each 1% increase in fibrosis, the risk of recurrence climbed by 6.3%. Among the factors adjusted for in this multivariate analysis were age, sex, hypertension, heart failure, mitral valve disease, diabetes, AF type, left atrial volume, and procedure type.
Dr. Richard I. Fogel, chair of the scientific sessions program committee and HRS president-elect, commented that he found it surprising and counterintuitive that AF duration was not a predictor of the extent of atrial fibrosis in DECAAF.
"I thought that the theory was ‘atrial fibrillation begets atrial fibrillation,’ " said Dr. Fogel, a cardiologist and electrophysiologist in group practice in Indianapolis.
Dr. Marrouche replied that large gaps remain in understanding the pathophysiology of AF in humans.
"We’ve known for a long time from animal models that AF begets AF. With MRI, for the first time we’re opening the heart in humans with AF, and it doesn’t look like the animal models at all. The fibrosis in humans isn’t diffuse; it’s patchy, and it’s localized," according to Dr. Marrouche.






