Development of Primary Cutaneous Anaplastic Large Cell Lymphoma Following Treatment With Upadacitinib for Atopic Dermatitis
Practice Points
- Janus kinase inhibitors are immunomodulators used for the treatment of various inflammatory conditions, including atopic dermatitis.
- Treatment with Janus kinase inhibitors may be associated with the development of CD3012+ lymphoproliferative disorders such as cutaneous anaplastic large cell lymphoma.
To the Editor:
A 22-year-old man presented to our clinic with a history of longstanding widespread recalcitrant atopic dermatitis (AD) since early childhood. He had been treated by an outside physician with topical steroids and nonsteroidal medications without notable improvement as well as with dupilumab, which was discontinued due to the development of severe head and neck dermatitis. Given the severity of his AD on presentation, we initiated treatment with upadacitinib 15 mg/d, which resulted in partial improvement. The dose was increased to 30 mg/d at 3 months with further clinical improvement.
Ten months after the patient was started on upadacitinib, he presented for a follow-up evaluation and reported a new nontender nodule on the scalp. A punch biopsy revealed a dense dermal and subcutaneous lymphoid infiltrate (Figure 1) composed of many large atypical CD2+/CD5+/CD45+ T cells with partial loss of CD3 expression (Figure 2). The atypical cells demonstrated diffuse CD30+ expression (Figure 3) and a CD4:CD8 ratio of greater than 50:1 (Figures 4 and 5). He was diagnosed with anaplastic large cell lymphoma (ALCL), and the upadacitinib was discontinued. No additional therapies directed toward ALCL were initiated.
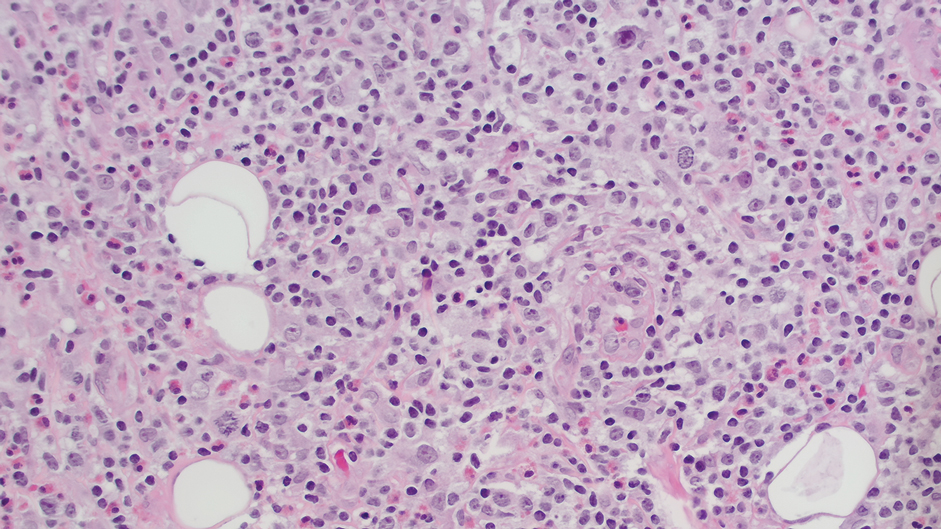
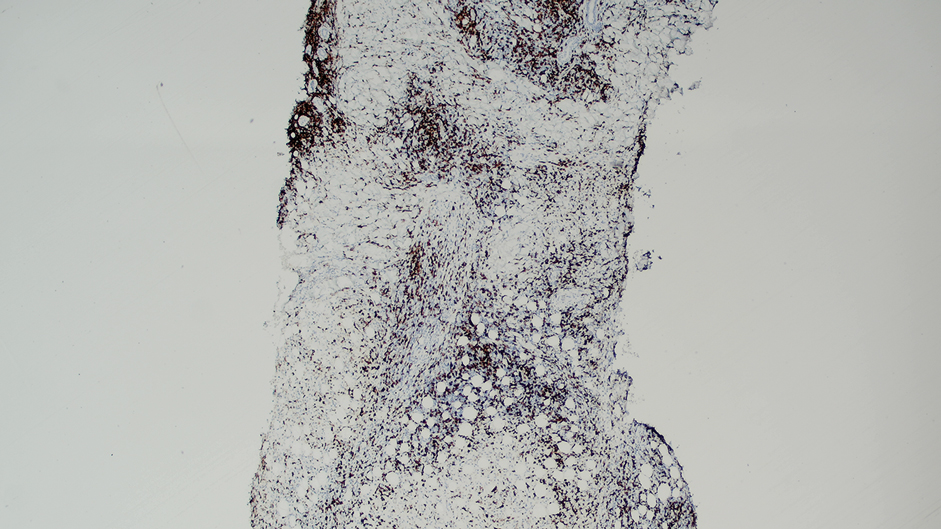
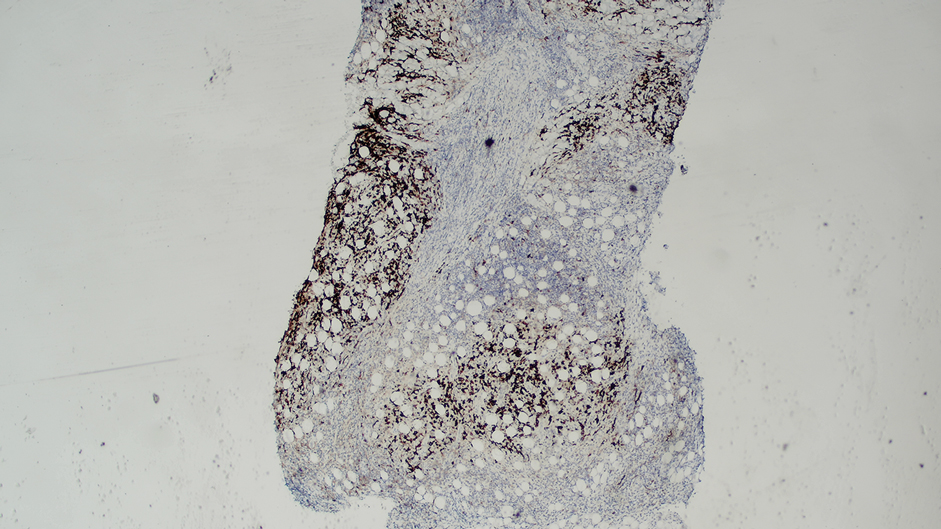
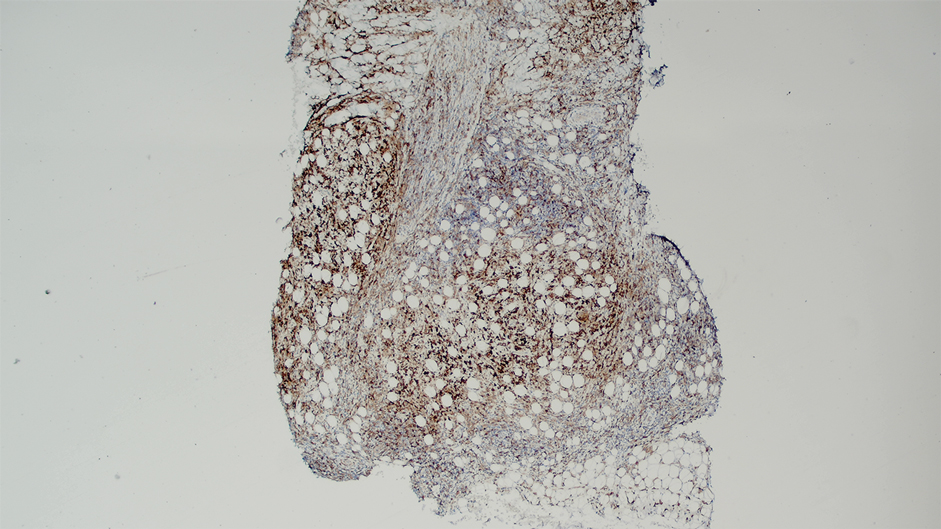
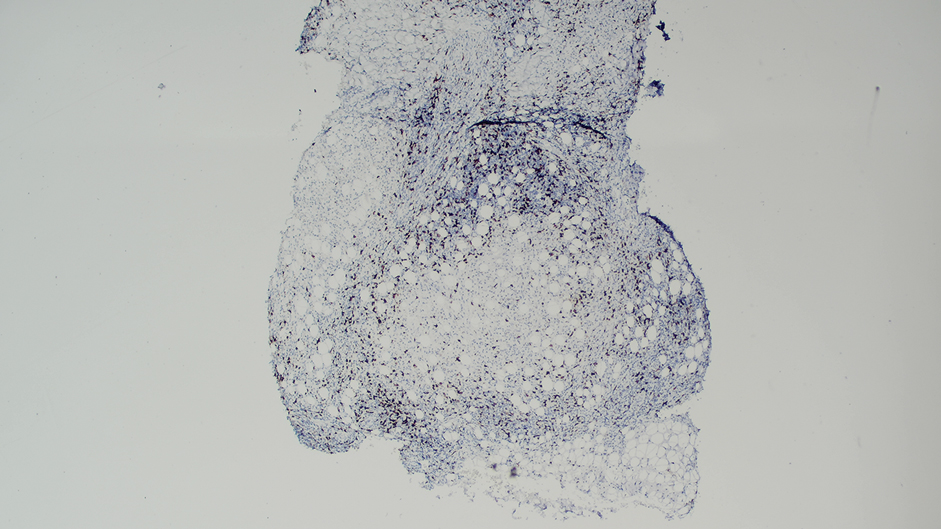
Over the next 2 weeks, the patient developed additional nodules on the postauricular skin and trunk that demonstrated similar histopathology and immunophenotype to the original scalp nodule. T-cell receptor gene rearrangement studies demonstrated shared clonal peaks in these subsequent nodules. A concurrent biopsy of an eczematous plaque on the back showed spongiotic dermatitis without evidence of cutaneous T-cell lymphoma; gene rearrangement studies from this site were negative. A positron emission tomography–computed tomography scan showed mildly hypermetabolic cervical, axillary, and inguinal lymph nodes, which were favored to be reactive. Narrow-band UVB phototherapy was initiated for management of the AD, and no additional nodules developed over the subsequent months.
Janus kinase (JAK) inhibitors are immunomodulatory small molecules that interfere with JAK–signal transducer and activator of transcription signaling involving 1 or more isoforms (eg, JAK1, JAK2, JAK3, tyrosine kinase 2) and have been used to treat various inflammatory conditions, including rheumatoid arthritis, psoriatic arthritis, psoriasis, axial spondyloarthritis, inflammatory bowel disease, and AD.1 Upadacitinib is an oral selective JAK1 inhibitor approved by the US Food and Drug Administration for treatment of moderate to severe AD in adults and children aged 12 years and older.2 A search of PubMed using the terms upadacitinib or Rinvoq and anaplastic large cell lymphoma did not identify any cases of cutaneous ALCL arising after treatment with upadacitinib. However, a case of lymphomatoid papulosis after initiation of upadacitinib for the treatment of rheumatoid arthritis in a 74-year-old Japanese woman has been described,3 and the JAK/signal transducer and activator of transcription pathway has been implicated in the development of other CD30+ lymphoproliferative disorders.4,5
An association between JAK inhibitors and aggressive B-cell lymphomas has been described. In an observational study of 626 patients with myeloproliferative neoplasia by Porpaczy et al,6 4 of 69 (5.8%) patients treated with JAK inhibitors developed an aggressive B-cell lymphoma, whereas only 2 of 557 (0.36%) patients who did not receive JAK-inhibitor therapy developed an aggressive B-cell lymphoma. In contrast, a retrospective analysis of 2583 patients with myeloproliferative neoplasia by Pemmaraju et al7 found no significant increase in lymphoma rates in the JAK inhibitor–treated population as compared with the non-JAK inhibitor–treated group; 9 (0.56%) cases of lymphoma occurred in 1617 patients with myelofibrosis, of which 6 had exposure to JAK inhibitor therapy and 3 had no exposure to JAK inhibitor therapy (P=.082) and 5 (0.52%) cases of lymphoma occurred in 966 patients with essential thrombocythemia or polycythemia vera, none of whom had exposure to JAK inhibitor therapy.Finally, some evidence suggests the use of JAK inhibitors may be associated with an elevated risk of malignancies overall. The ORAL Surveillance study found the incidence of all cancers, excluding nonmelanoma skin cancer (NMSC), in patients treated with tofacitinib to be 4.2% (122/2911) compared with 2.9% (42/1451) in patients treated with tumor necrosis factor α inhibitors; it should be noted that the patients in this study were restricted to adults aged 50 years and older who were undergoing treatment for rheumatoid arthritis.8 In a safety profile study for upadacitinib, a higher rate of malignancies, excluding NMSC, was found in patients with AD treated with upadacitinib 30 mg/d than in patients treated with 15 mg/d; however, the overall rates of malignancies, excluding NMSC, in patients treated with upadacitinib were comparable to the standard incidence rates of malignancies in the general population derived from Surveillance, Epidemiology, and End Results data.9
In summary, we present a case of cutaneous ALCL arising after treatment with upadacitinib for AD. While some literature suggests AD may independently predispose patients to the development of CD30+ lymphoproliferative disorders, the onset of our patient’s cutaneous ALCL 10 months after initiation of upadacitinib is suggestive of an association between his lymphoproliferative disorder and JAK inhibition. Further studies are needed to better characterize the risk of lymphoproliferative disorders and other malignancies in patients treated with JAK inhibitors.




