Thermal Therapy is Associated With Decreased Pain Treatment Use: A Retrospective Cohort Study
Background: Body surface heat and cold (eg, thermal) therapies are nonpharmacologic pain treatments purported to have pain-reducing benefits, but there is limited research on their use for chronic pain. The purpose of this study was to examine the potential clinical and health system benefits of thermal therapy.
Methods: Participants included 2182 Veterans Health Administration patients who received a thermal therapy device and 2182 propensity score-matched control patients. Clinical encounters, pain pharmacy data, and pain self-ratings were extracted from the electronic health record in the year prior to and following receipt of a device, or the corresponding dates for matched control patients. Mixed-effects regression and random-effects growth modeling were used to compare changes in pain treatment utilization and pain intensity, between thermal therapy recipients and matched controls.
Results: Patients who received thermal therapy had greater reductions in days of high-dose opioid therapy use (Time × Treatment interaction, B = 3.24; 99% CI, 0.34 to 6.14; P < .01) and concurrent use of opioids and benzodiazepines (Time × Treatment interaction, incidence rate ratio [IRR], 0.76; 99% CI, 0.67 to 0.86; P < .01) in the year following device receipt when compared with matched control patients. In addition, patients who received thermal therapy had fewer encounters with pain specialty care, including physical therapy (Time x Treatment interaction, IRR, 1.16; 99% CI, 1.00 to 1.33; P = .01) and pain clinics (Time x Treatment interaction, IRR, 1.49; 99% CI, 1.22 to 1.76; P < .01). Mean pain intensity remained relatively stable for both groups (B = -0.02; 99% CI, -0.06 to 0.02; P = .15).
Conclusions: While associations identified in this observational study do not establish causality, thermal therapy may confer health system benefits through decreased use of costly specialty pain care and high-risk opioid use.
Growing recognition of the limitations of long-term opioid therapy for chronic noncancer pain has highlighted the importance of nonpharmacologic approaches to pain treatment.1,2 These treatments are varied and may include psychological and behavioral therapies (eg, cognitive behavioral therapy for chronic pain), exercise and movement therapies (eg, yoga), and manual therapies (eg, chiropractic). Body surface cold therapy, while predominantly used to reduce postoperative pain and inflammation,3,4 pain subsequent to acute musculoskeletal injury,5 and pain related to muscle soreness,6 is a nonpharmacologic treatment that has shown pain-reducing benefits for chronic low back pain and knee osteoarthritis, as has its counterpart, superficial heat therapy.7-9 Heat therapy has also been shown to improve strength, flexibility, and activities of daily living in patients with chronic low back pain.10,11 Cold and heat therapies are commonly used complementarily. Cold therapies aim to reduce blood flow and inflammation and are often used immediately following trauma to an affected area, whereas heat therapies increase blood flow and metabolic activity and are commonly used to promote healing.5
Heat and cold therapies (also known as thermal therapies) benefit resource-limited health care systems, as most devices require a single expenditure and can be self-administered by patients at home as part of their pain self-management plan. In addition, these pain self-management tools may attenuate the need for more expensive specialty pain care and ongoing analgesic pharmacotherapy. Despite their potential, few studies have characterized the benefits of thermal therapies for patients with heterogeneous chronic pain syndromes.
The purpose of this retrospective cohort study was to examine the potential clinical and health system benefits of patient-administered thermal therapy. Our primary hypothesis was that patients with chronic musculoskeletal pain who received a thermal therapy self-management device would have fewer days of opioid prescriptions compared with a sample of matched control patients. Secondarily, we hypothesized that patients who received a thermal therapy device would have lower utilization of specialty pain care, fewer potentially hazardous opioid prescriptions (eg, high-dose opioid therapy and concurrent opioid and benzodiazepine prescriptions), fewer prescriptions for nonopioid analgesic medications, and decreased pain intensity when compared with matched controls.
Methods
This retrospective cohort study compared pain pharmacotherapy, pain treatment utilization, and pain intensity outcomes between patients who received a thermal therapy device and matched patients who did not. The study was approved by the US Department of Veterans Affairs (VA) Portland Health Care System Institutional Review Board and was granted a waiver of informed consent to access patient electronic health records (EHRs).
Pain Care
The VA uses ThermaZone thermal therapy devices (Innovative Medical Equipment) for chronic pain treatment. The device uses thermoelectric technology to provide point-of-contact cooling and heating therapy through site-specific pads (eg, ankle, knee, hip, back, elbow, shoulder). Patients place pads on sites where they experience pain, and temperature regulated water circulates through the device and to the pad, providing consistent, localized thermal therapy. The pads range in temperature from 1 °C to 52 °C, and temperatures are self-monitored and controlled by the patient.
Standard pain care in this study followed the VA stepped model of pain care, which builds on a foundation of patient education for pain self-management approaches (eg, exercise, mindfulness, relaxation, social support).12 According to the VA stepped model of pain care, all patients with chronic pain should engage in these foundational self-management approaches. However, some patients require more intensive care. The VA stepped-up treatment engages primary and specialty care services—such as physical therapy, pharmacy, complementary and integrative health approaches, mental health, and substance use services—and, when indicated, may escalate treatment to interdisciplinary pain teams or tertiary pain centers. In this retrospective cohort study, treatment patients received a thermal therapy device and standard of care, whereas control patients received standard care only.
Sample Selection
Eligible patients were aged ≥ 18 years, had a musculoskeletal pain diagnosis documented in the EHR in the year prior to thermal therapy device receipt (or during the same period for a treatment patient’s matched control), and were enrolled in VA health care during the study period. Patients who died during the study period were excluded. Treatment patients received a thermal therapy device from the VA between January 1, 2017, and December 31, 2018, when complete data on thermal therapy devices were available. For control patients, the VA Corporate Data Warehouse (CDW) was used to identify VA patients with characteristics similar to those of treated patients.
We modeled the probability that a patient would receive a thermal therapy device using logistic regression. Predictor variables were measured in the year prior to device receipt and included variables associated with pain treatment utilization and analgesic pharmacotherapy receipt, as recommended by Brookhart et al.13 These included age, sex, race, ethnicity, VA service-connected disability status, comorbidities, receipt of medications for opioid use disorder, pain diagnoses, mental health diagnoses, and substance use disorder diagnoses.14-19
The resulting propensity scores (eg, predicted probabilities) were used to match treatment patients 1:1 with control patients using a nearest-neighbor matching algorithm.20 This procedure matches a control patient with the closest propensity score to that of the corresponding treatment patient. An exact match on VA facility was required (eg, treatment patients and matched controls received care at the same VA facility). Standardized differences were used to assess covariate balance between the matched groups, and kernel density plots of propensity scores tested for sufficient overlap.21 Control patients were selected from a pool of 1,150,149 patients.
Study Variables
The index date was the date the thermal therapy device was released to treatment patients or the same date for the matched controls. Data were extracted from the CDW over a 24-month period: 12 months prior to the index date through 12 months afterwards. Collecting data in the 12 months prior to treatment initiation allowed us to adjust for covariates and provided greater precision, as recommended for observational study designs.22
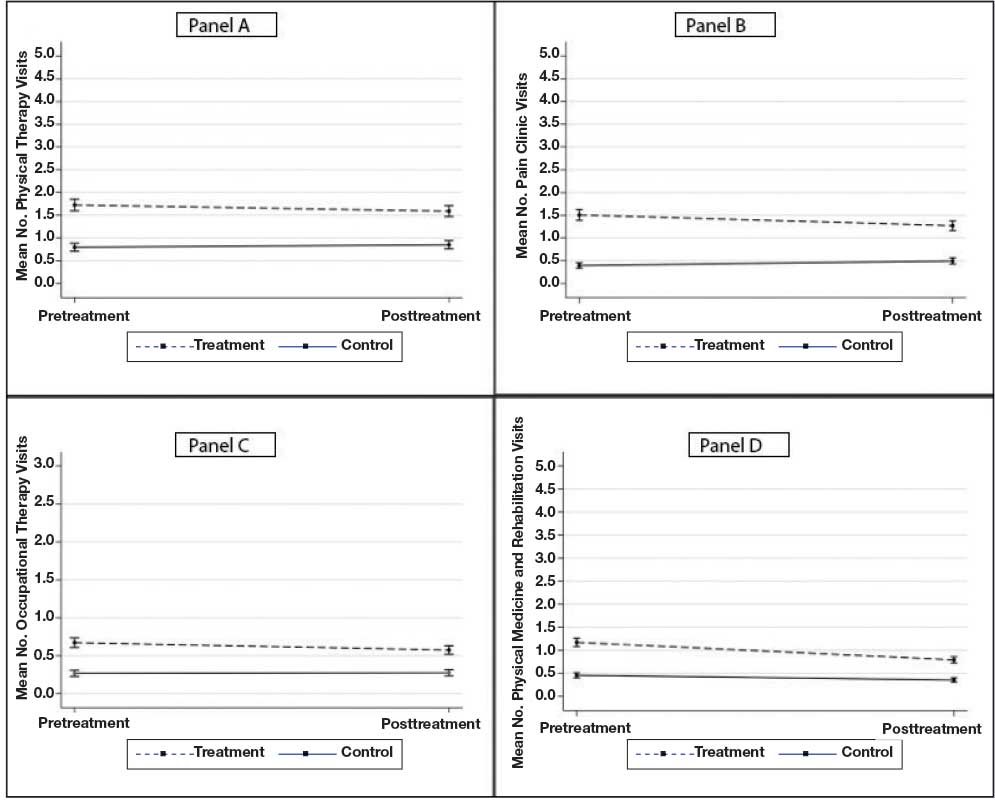
Treatment conditions were defined dichotomously as receipt vs nonreceipt of a thermal therapy device. The primary outcome was the number of days of opioid use in the 12 months following the index date. Additional outcomes included days of high-dose opioid therapy (≥ 50 mg morphine equivalent [MME] daily), concurrent opioid and benzodiazepine prescriptions, and nonopioid analgesic pharmacotherapy (eg, nonsteroidal anti-inflammatory drugs, acetaminophen, muscle relaxants). All prescription data were based on medication fills obtained from VA pharmacy records. Additional outcomes included the number of visits to physical therapy, occupational therapy, physical medicine and rehabilitation, and interdisciplinary pain clinics, including interventional pain medicine.
Pain intensity ratings were collected as part of routine VA care using a numeric scale from 0 (no pain) to 10 (worst possible pain). Pain intensity trajectories were computed using all available EHR-derived pain intensity score data for each patient in the 12 months prior to and following the index date.
Covariates were extracted from the EHR and evaluated in the year prior to the index date, unless otherwise noted. They included age at the index date; self-reported sex, and race and ethnicity; service-connected disability status (disability awarded as a result of military service-related trauma or injury); Charlson Comorbidity Index; and diagnoses of opioid use disorder, alcohol use disorder, other substance use disorder, mood disorder, posttraumatic stress disorder, other anxiety disorder, psychotic disorder, neuropathic pain, and headache pain.23 All diagnoses were coded as yes if identified in the EHR as a focus of treatment during any clinical encounter in the year prior to the index date or no if not identified as a focus of treatment.
The number of days patients had been living with pain was calculated and defined as the number of days from the first pain diagnosis available in a patient’s EHR to the index date. Finally, the number of pain-related surgeries (eg, surgeries with ≥ 1 pain diagnoses associated with the clinical encounter) and average pain intensity were computed for the pre- and postindex date evaluation periods and included as model covariates.
Statistical Analyses
For the 4 pharmacotherapy and 4 nonpharmacologic treatment usage outcomes, we tested the fit of linear models and several models with count distributions using the Bayesian information criterion.24 Count distributions included Poisson, zero-inflated Poisson, negative binomial, and zero-inflated negative binomial. With 1 exception (days of high-dose opioid use), a negative binomial distribution best fit the data. For days of high-dose opioid use, a Gaussian distribution best fit the data.
Eight separate mixed-effects regression analyses compared changes in each outcome from the 12-month preindex period through the 12-month postindex period between treatment and control patients by testing the Time × Treatment interaction. This approach statistically accounts for observed pretreatment differences in outcome variables. Statistics for the main effects of time and treatment are also presented. To reduce bias, models controlled for covariates specified previously.
For pain intensity, we used random-effects growth modeling to quantify both fixed and random effects of pain intensity at the index date (eg, the model intercept), which estimates pain at the time of treatment initiation, and change in pain during the 12 months following the index date (eg, the model slope), which characterizes the trajectory of pain intensity ratings.25 The model included piecewise components of pain score trajectories in the 12 months prior to the index date and in the 12 months following the index date. Several types of change for the 12-month postindex observation period were explored—including quadratic and cubic curvilinear change. A linear model for change in pain over time provided the best fit based on the Bayesian information criterion and parsimony of model parameters.26 We report estimates of change in pain over time in monthly intervals for ease of interpretation. However, models used all individual pain scores rather than computing monthly averages when > 1 pain score was available within a month, as recommended in previous research.27 This approach makes optimal use of all available data. Both random effects (intercept and slope) were regressed onto the set of covariates described previously.
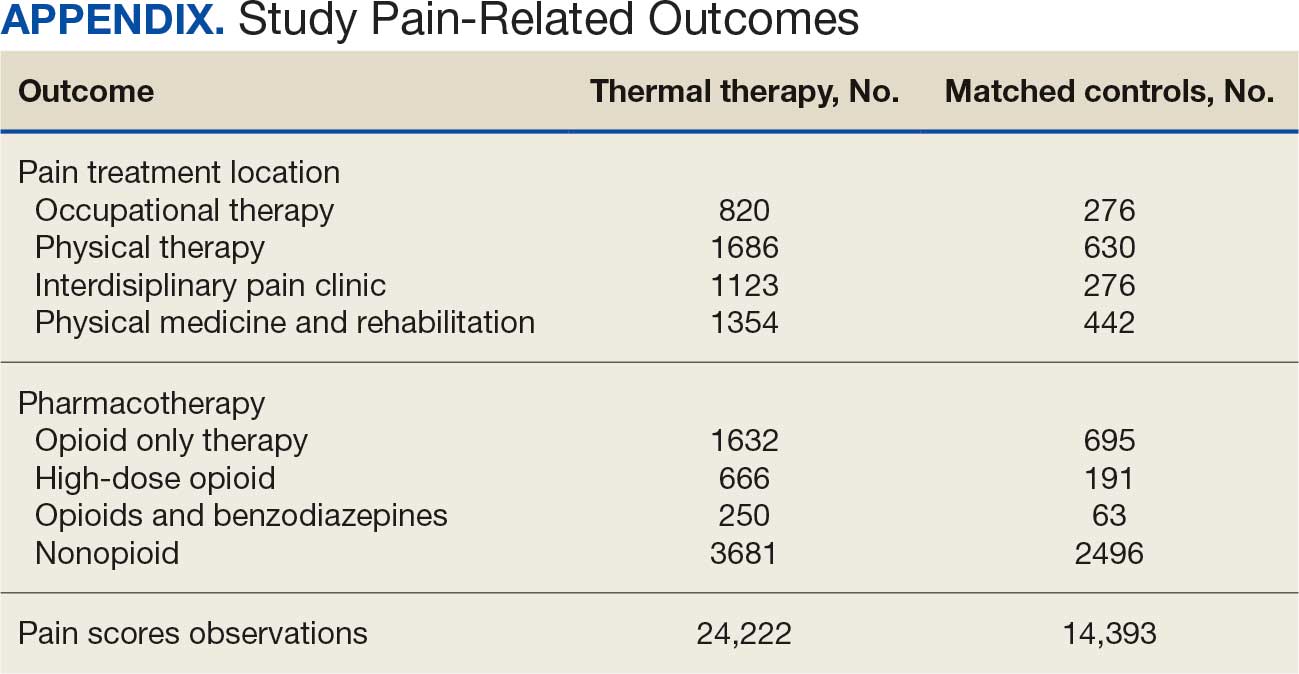
This study used data available in the EHR over the 24-month observation period. We characterize the density (eg, frequency) of all outcome variables by treatment condition in the Appendix. Because the hypotheses were directional, the authors used a 2-sided α = 0.10 and applied a Bonferroni correction for the 9 statistical tests performed, resulting in an adjusted α of 0.01. Treatment utilization and pharmacotherapy outcome analyses were performed in Stata, version 16.1. Random-effects growth modeling of pain score trajectories was performed using Mplus, version 8.8.
Results
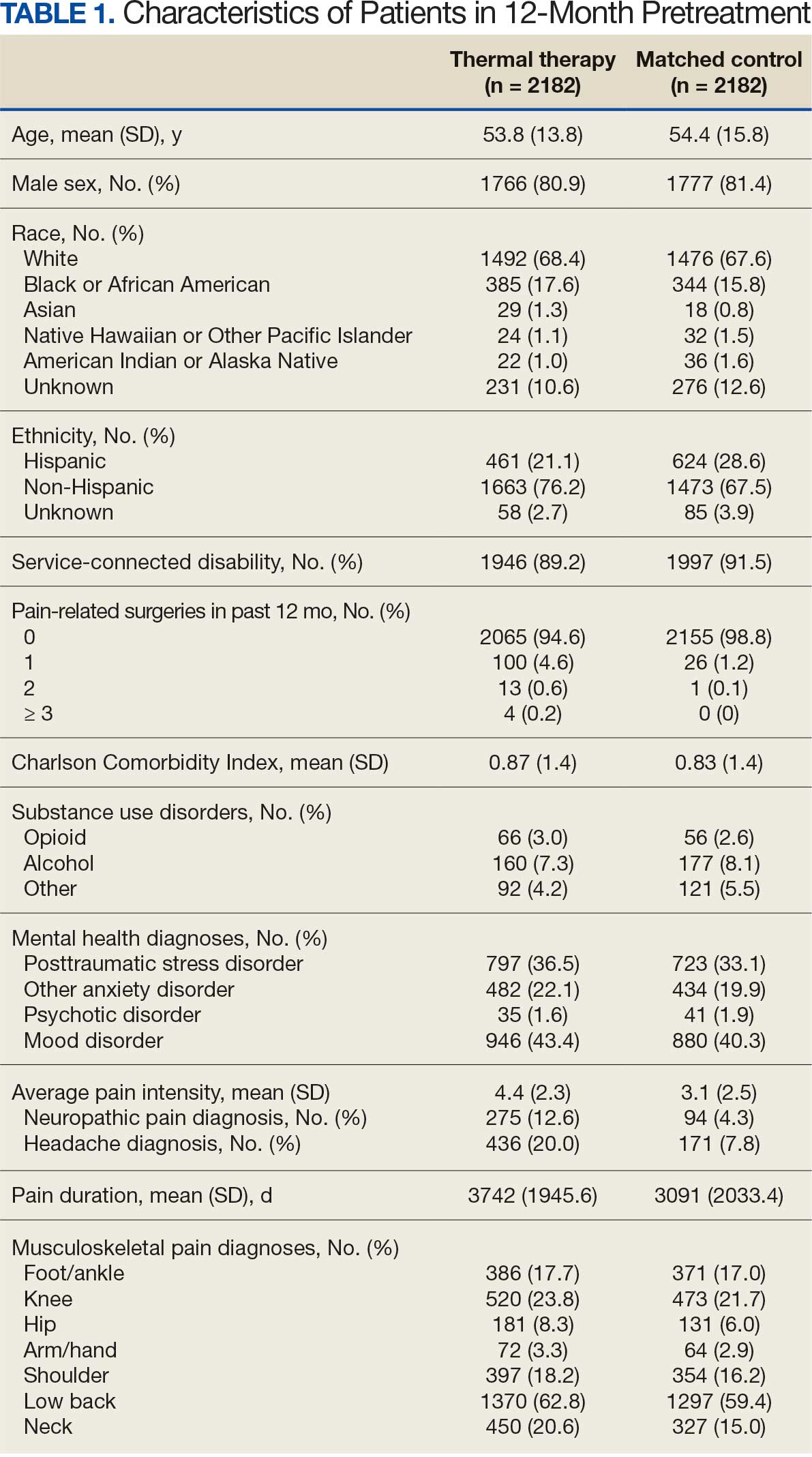
There were 2182 patients in the treatment group and 2182 matched controls. The mean (SD) age was 54 (15) years; 81% were male, and about two-thirds (68%) identified as White and non-Hispanic. Mental health comorbidities were common, with > 40% of the sample having diagnoses of a mood disorder and/or posttraumatic stress disorder. Nearly all patients (90%) had VA service-connected disability ratings. Among patients with chronic musculoskeletal pain, 8% had comorbid neuropathic pain and 14% had headache. The mean (SD) duration of chronic pain across both patient groups was 3416 (2016) days, or about 9.4 years (Table 1).
Pharmacotherapy
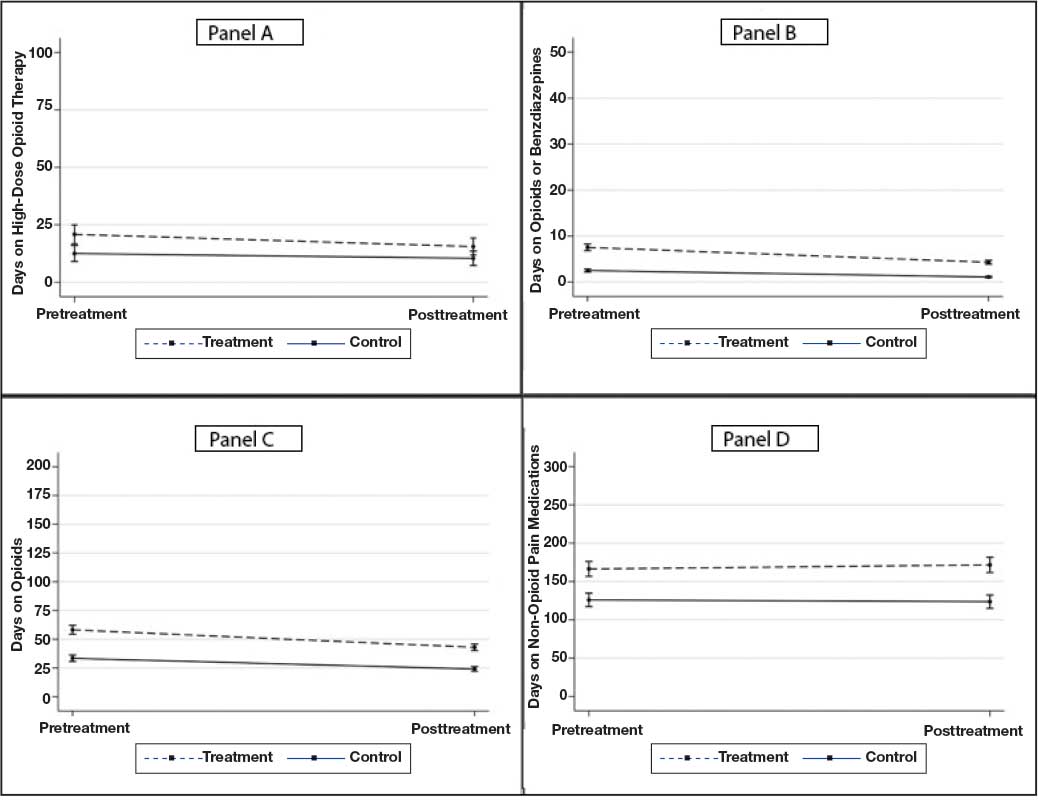
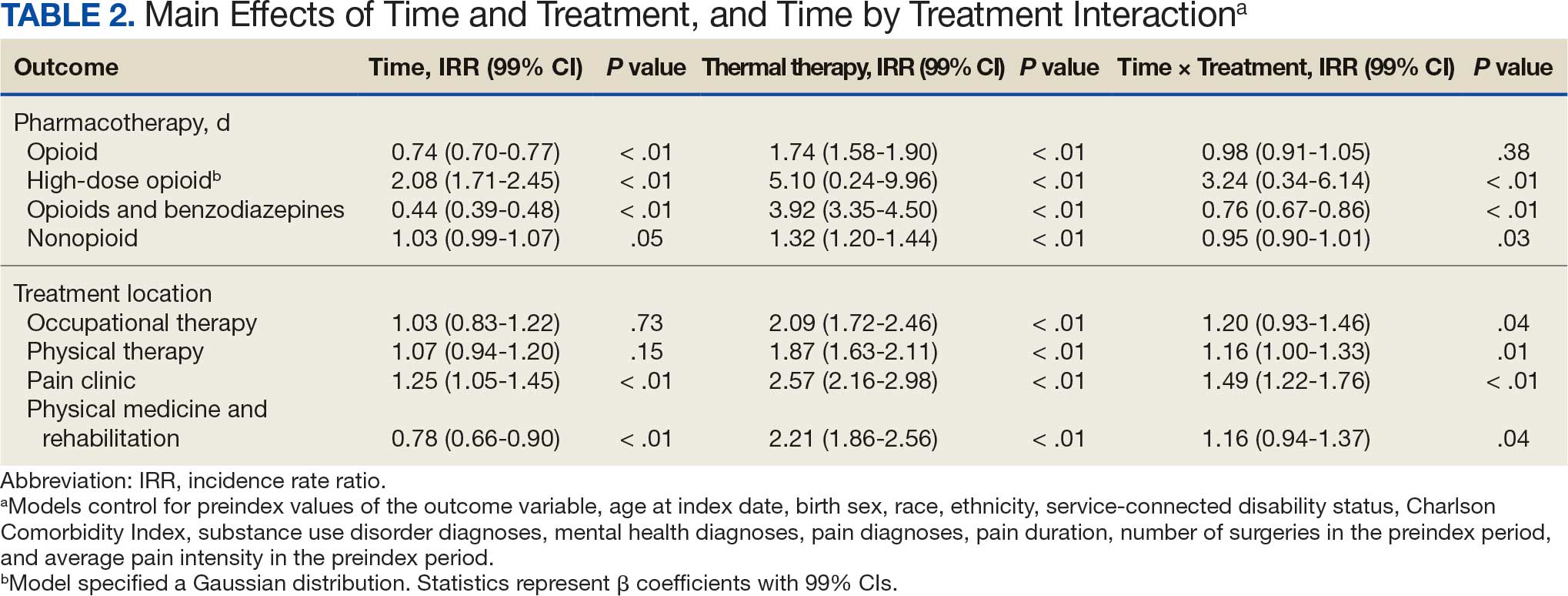
High-dose opioid use (> 50 MME daily dose), days of opioid use, and concurrent opioid and benzodiazepine use decreased for all patients from the pre- to posttreatment period (Table 2). However, high-dose opioid use (Time × Treatment interaction, 3.24; 99% CI, 0.34 to 6.14) (Figure 1A) and concurrent opioid and benzodiazepine use (Time × Treatment interaction, 0.76; 99% CI, 0.67 to 0.86) (Figure 1B) had a larger decrease for the treatment vs matched control group. Treatment and matched control patients had comparable reductions in days of opioid use (Time × Treatment interaction, 0.98; 99% CI, 0.91 to 1.05) (Figure 1C). Neither group showed changes in nonopioid analgesic pharmacotherapy over time (main effect of time, incidence rate ratio [IRR], 1.03; 99% CI, 0.99 to 1.07; Time × Treatment interaction, 0.95; 99% CI, 0.90 to 1.01) (Figure 1D).
Nonpharmacologic Pain Treatment
The number of physical therapy and pain clinic visits declined for treatment patients and increased slightly for matched control patients (Figure 2A and 2B). For occupational therapy visits, neither group showed changes over time (main effect of time IRR, 1.03; 99% CI, 0.83 to 1.22; Time × Treatment interaction, 1.20; 99% CI, 0.93 to 1.46) (Figure 2C). For physical medicine and rehabilitation visits, both groups decreased use over time (main effect of time IRR, 0.78; 99% CI, 0.66 to 0.90), but this decrease did not differ between treatment and control patients (Time × Treatment interaction, 1.16; 99% CI, 0.94 to 1.37) (Figure 2D).
Pain Intensity
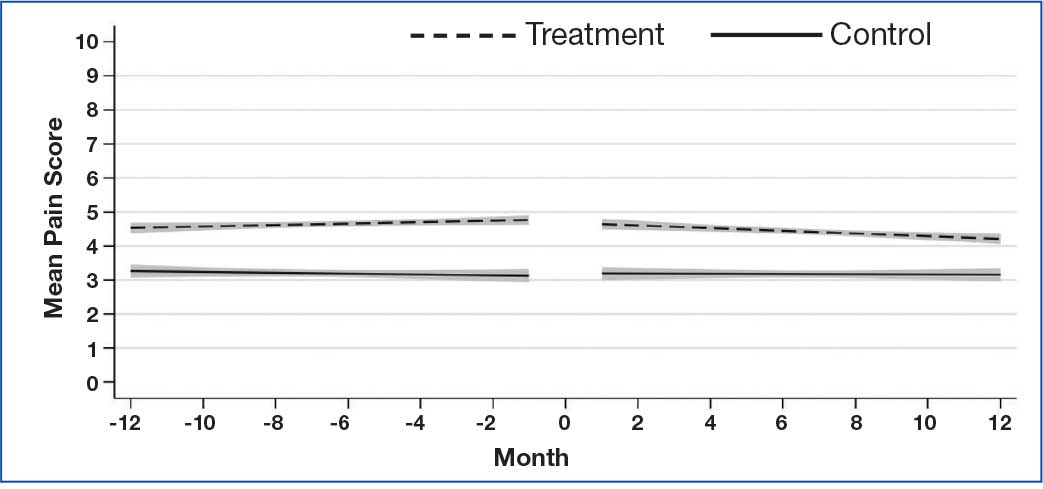
Pain intensity decreased for both groups by an estimated 0.02 points per month (99% CI, -0.04 to -0.01; P < .01), or 0.24 points over the 12-month postindex follow-up period (Figure 3). There were no statistically significant differences between treatment and control patients in pain intensity changes over the follow-up period (B = -0.02; 99% CI, -0.06 to 0.02; P = .15).
Discussion
Patients with musculoskeletal pain who received a thermal therapy device showed a larger decline in use of some specialty pain treatments, including physical therapy and specialty pain clinic services, when compared with matched control patients. One possible explanation is that patients who used the device may have had their pain adequately managed and thus required less specialty pain care. However, the absence of clinically significant changes in pain intensity over time suggests that pain intensity alone did not account for the observed changes in pain treatment use. We were unable to measure constructs of pain interference or functioning, which may be better predictors of functional restoration, as these data are not routinely collected within the VA and documented in the EHR. Future studies could clarify these findings by including measures of pain interference and functioning.
Although the overall declines in physical therapy and specialty pain clinic use associated with thermal therapy were modest (< 1 visit per patient), the impact of these reductions can be profound. In resource-limited health care settings, even small reductions in high-cost care utilization could be of great value in that health systems could offset costs associated with treating chronic pain without compromising quality of care or key clinical outcomes, such as pain intensity. This study, however, did not include a cost analysis. Future studies should incorporate formal cost analyses to quantify cost offsets that may result from decreased specialty pain treatment use.
Patients who received thermal therapy devices did not show clinically meaningful reductions in pain intensity over time, defined as reductions in pain intensity of 1.7 to 2.0 on a 0 to 10 scale.28,29 This finding is consistent with prior research that demonstrates relatively stable pain intensity self-ratings longitudinally by patients with chronic pain diagnoses, when assessed in the context of usual clinical care.30 This finding, however, is inconsistent with prior literature that demonstrates pain-reducing benefits of thermal therapy for low back pain and knee osteoarthritis.7-9
In this study, pain intensity ratings were derived from the EHR during routine outpatient clinical encounters and not at the time thermal therapy was self-administered, as has been done in prior clinical trials.7-9 Pain location was not specified at the time of pain ratings, and it is possible that patients may have been endorsing pain in areas of the body that had not been treated by thermal therapy. Patient-level variability in pain intensity ratings (eg, within-patient range over time) was not examined, although prior research indicates substantial variability.30 While average pain intensity ratings in the current study did not change, an examination of patient variability warrants further study, as a narrowing of pain intensity ratings can be perceived, by patients, as demonstrable improvement and has been associated with improved physical and psychological outcomes.31 Furthermore, pain intensity does not characterize physical or emotional functioning that can be captured with more comprehensive validated measures, some of which are recommended outcomes in pain clinical trials.32
Our findings point to reductions in all forms of opioid use across both treatment and control patients. Data from the VA and in the US more generally point to downward trends in opioid prescribing during the study period.33 This decline is likely due to increased use of risk mitigation approaches, such as routine urine drug screens and review of prescription drug monitoring databases.34 These state-level databases track prescribing of controlled substances, including opioids and benzodiazepines, within a state. Implementation of these practices has been associated with declines in higher risk opioid prescribing.34 Findings from this study further point to associations of reduced higher risk opioid use among patients who received thermal therapy devices. In the full sample of patients, reductions in days of opioid use, high-dose opioid use, and co-use of opioids and benzodiazepines were observed across all patients, with greater reductions observed in high-dose opioid use and concurrent opioid and benzodiazepine use among patients who received a thermal therapy device. Experimental studies indicate that the endogenous opioid system is implicated in pain relief and activated by thermal therapies. 35 Differential reductions in higher-risk opioid use among patients who received thermal therapy devices in our study may be associated with endogenous opioid activation, though this was not specifically measured. It is also unclear whether thermal therapy was provided by clinicians in the context of opioid tapering or other risk mitigation efforts, or patients reduced higher risk opioid use of their own volition. Prior research has identified both patient- and clinician-initiated opioid tapering and discontinuation.36 While a thorough explication of opioid dose reduction was beyond the scope of this study, future qualitative work could help explain potential benefits of thermal therapy in the context of analgesic pharmacotherapy use, including opioid medications.
Limitations
The extent to which patients used the thermal therapy device could not be measured; therefore, device receipt was used as a proxy for use. However, it was not possible to determine whether the frequency and duration of device use was associated with study outcomes. Treatment and control groups demonstrated some differences in outcome variables at the index date. Potential known biases were addressed using propensity score matching procedures and statistical procedures that controlled for patient demographic and clinical characteristics, as well as pretreatment values of all outcome variables. Nevertheless, as an observational study, this analysis cannot account for all known and unknown confounders, and a randomized controlled trial is needed to make claims of causality. The study population consisted of US veterans and included a low proportion of women. As a result, the findings may not be generalizable to other patient populations. Finally, prescription dispensing data, used as a proxy for pharmacotherapy use, do not necessarily reflect actual medication use.
Conclusions
This study is among the first to examine associations between thermal therapy and specialty pain treatment and analgesic pharmacotherapy use among US veterans. Although the retrospective cohort study design does not allow causal inferences regarding the efficacy of thermal therapy for veterans with chronic musculoskeletal pain, confidence in the findings is strengthened by methodological and statistical control of known confounders. Future trials employing experimental designs are needed to further clarify the clinical and health systems benefits of thermal therapy for musculoskeletal pain syndromes.




