UPDATE: ENDOMETRIAL CANCER
Are lymphadenectomy and external-beam radiotherapy valuable in women who have an endometrial malignancy? Debate over the standard of care continues.
IN THIS ARTICLE
Dr. Mutch reports that he has received grant or research support from Lilly and Genentech. He serves as a speaker for GSK, Lilly, and Merck. Dr. Rimel reports no financial relationships relevant to this article.
Endometrial cancer is a great concern in industrialized nations, where it is the most common gynecologic cancer—with incidence increasing every year. Survival is generally very good for women who have low-grade disease confined to the uterus. However, for patients who have high-grade disease, an aggressive histologic type, or other features that suggest a poor prognosis, the cure rate approaches 75%.1
Primary surgery is the mainstay of initial treatment and basis of FIGO staging ( TABLE ), which requires:
- total hysterectomy
- bilateral salpingo-oophorectomy
- complete examination of the abdomen
- pelvic washings
- lymph-adenectomy (anatomic boundaries and node counts aren’t specified).
Controversy clouds our understanding of the optimal type of surgery, utility of pelvic lymphadenectomy, and possible benefit of adjuvant radiation therapy. During the past year, fuel has been added to this debate:
- Two randomized, controlled trials of surgery with and without pelvic lymphadenectomy in early-stage patients demonstrated no survival benefit. Earlier studies investigating the benefits of lymphadenectomy in endometrial cancer have been largely retrospective, and results have varied.
- A concurrent randomized, controlled trial of external-beam radiotherapy for women who have intermediate- or high-risk disease showed no improvement in overall survival, although local control increased by 3%.
TABLE
FIGO surgical staging for endometrial cancer
| Stage | Description |
|---|---|
| I | Tumor is confined to uterine fundus |
| IA | Tumor is limited to endometrium |
| IB | Tumor invades less than half of the myometrial thickness |
| IC | Tumor invades more than half of the myometrial thickness |
| II | Tumor extends to cervix |
| IIA | Cervical extension is limited to endocervical glands |
| IIB | Tumor invades cervical stroma |
| III | There is regional tumor spread |
| IIIA | Tumor invades uterine serosa or adnexa, or cells in the peritoneum show signs of cancer |
| IIIB | Vaginal metastases are present |
| IIIC | Tumor has spread to lymph nodes near the uterus |
| IV | There is bulky pelvic disease or distant spread |
| IVA | Tumor has spread to bladder or rectum |
| IVB | Distant metastases are present |
No survival advantage to pelvic lymphadenectomy—but it has other benefits
ASTEC study group, Kitchener H, Swart AM, Qian Q, Amos C, Parmar MK. Efficacy of systematic pelvic lymphadenectomy in endometrial cancer (MRC ASTEC trial): a randomised study. Lancet. 2009;373:125–136.
Benedetti Panici P, Basile S, Maneschi F, et al. Systematic pelvic lymphadenectomy vs. no lymphadenectomy in early-stage endometrial carcinoma: randomized clinical trial. J Natl Cancer Inst. 2008;100:1707–1716.
Among the arguments for lymphadenectomy in endometrial cancer staging are:
- It aids in the selection of women for radiation or other adjuvant treatment
- It may have a direct survival benefit, as suggested by retrospective studies.
But lymphadenectomy is time-consuming, requires a specialized gynecologic surgeon, and is associated with some increase in the risk of morbidity—namely, lymphedema, lymphocyst formation, deep-vein thrombosis (DVT), and blood loss.
The much-anticipated report of the 85-center, multinational ASTEC trial [ A S urgical T rial of E ndometrial C ancer], published earlier this year, offers further insight into the practice of lymphadenectomy. ASTEC involved two randomizations: The first, to pelvic lymphadenectomy; the second, to radiation therapy.
The ASTEC trial enrolled 1,408 women who had histologically confirmed endometrial carcinoma that was believed to be confined to the uterus. How this determination was made was not specified. Patients who had enlarged lymph nodes corroborated by computed tomography or magnetic resonance imaging were not excluded.
Participants were randomized to either of the following treatment groups:
- traditional surgery with total hysterectomy and bilateral salpingo-oophorectomy, pelvic washings, and palpation of para-aortic nodes
- the same surgery plus systematic lymphadenectomy of the iliac and obturator nodes.
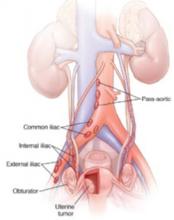
If any para-aortic nodes were suspicious, biopsy or lymphadenectomy was performed at the discretion of the surgeon ( FIGURE ).
FIGURE Nodes reveal when cancer has spread
Women in the ASTEC trial were randomized to traditional surgery (total hysterectomy and bilateral salpingo-oophorectomy), pelvic washings, and palpation of para-aortic nodes or to the same surgery plus lymphadenectomy of the iliac and obturator nodes.
Operative findings determined a patient’s level of risk
After surgery, patients were categorized as having one of the following:
- low-risk, early-stage disease. This group included patients who had disease classified as stage IA or IB, grade 1 or 2. They were deemed to have a suitably low risk of recurrence to be offered further treatment according to their physician’s standard practice.
- intermediate- or high-risk, early-stage disease. These patients were randomized to the ASTEC radiation-therapy trial, which compared external-beam radiotherapy with no external-beam radiotherapy. The authors assert that this second randomization was necessary to prevent over- or undertreatment of patients who had unknown node status, which might alter survival outcomes.
- advanced disease. These patients were referred to their physician for further treatment.