Q Does Botox relieve urinary urgency and urge incontinence?
<huc>A</huc> Yes, but improvements are time-limited. In this series of 100 cases of idiopathic detrusor overactivity treated with botulinum toxin A (Botox), symptoms resolved in 74% of patients with urge incontinence by 4 weeks after treatment, and in 80% of patients by 12 weeks after treatment. Urgency resolved in 72% at 4 weeks and 66% at 12 weeks.
Expert Commentary
This fairly large case series helps clarify the potential of Botox to ease symptoms in patients who do not respond to current therapies for idiopathic detrusor overactivity.
Among the first authors to report successful use of Botox for neurogenic detrusor overactivity was a research team from Switzerland led by Brigitte Schurch—the same group that published this case series. Participants met detailed criteria for “overactive bladder syndrome,” as defined by the International Continence Society, with either (1) urodynamically demonstrated nonneurogenic (idiopathic) detrusor overactivity with or without incontinence or (2) hypersensitive bladder with premature filling sensation (even when maximum bladder capacity was normal) and more than 8 voids per 24 hours (urgency–frequency syndrome with or without incontinence).
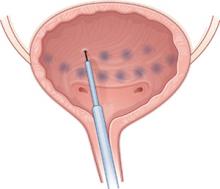
Participants included 77 women and 23 men, each of whom received 100 U of Botox diluted with saline and injected at 30 sites in the bladder under cystoscopic guidance, sparing the trigone.
Risks versus benefits
Adverse events reported in this study included urinary tract infections in 10 patients (10%) and urinary retention requiring intermittent self-catheterization in 4 patients (4%). However, retention was otherwise not well characterized. Mean postvoid residual did not return to baseline values until 9 months after injection.
Why these data are imperfect
Although the study included both women and men (ratio about 3:1), data were not reported by sex. Moreover, according to the International Continence Society definition, patients with overactive bladder syndrome constitute a heterogeneous group based on urinary symptoms (which may or may not include urge incontinence) and urodynamic findings (which may or may not include detrusor overactivity).
A small percentage of patients had apparent detrusor hypocontractility or acontractility with elevated postvoid residual urine volumes—a group clinically distinct from patients with otherwise “normal” bladder emptying despite symptoms attributed to the detrusor muscle.
A robust placebo effect is likely
In this open-label series, as in clinical practice, both patients and clinicians expected treatment to be beneficial—raising the possibility of a placebo effect that may explain part (or most) of the improvement. In many placebo-controlled drug trials involving patients with detrusor overactivity, a relatively large portion of the “treatment effect” may be attributed to the placebo effect; as many as 40% of patients taking placebo report some relief of symptoms.
Beyond beauty: Botox for the bladder
Under cystoscopic guidance, Schmid et al injected 100 U of saline-diluted Botox (tinted with indigo carmine to aid in spacing) at 30 sites in the bladder, excluding the trigone. Most women reported temporary relief of symptoms.
More data are needed
To address these gaps in knowledge, especially as use of Botox is being rapidly incorporated into clinical practice, investigators in the NIH-sponsored Pelvic Floor Disorders Network are conducting a randomized, placebo-controlled trial in women with idiopathic detrusor overactivity incontinence. The aim of the trial: to describe the percentage of women whose symptoms resolve, followed by time to recurrence, after treatment with Botox versus placebo, with 1 year of follow-up.
To obtain focused data, inclusion and exclusion criteria were developed to ensure that the population of this triple-blinded study is relatively homogeneous. As the occurrence and consequences of urinary retention have not been well described in prior studies, subjects in the NIH trial are being carefully assessed for urinary retention. Enrollment should be complete by mid-2007, and follow-up by mid-2008.
Should ObGyns consider Botox for women with intractable symptoms?
For clinicians experienced with cystoscopically guided injections, Botox might be a reasonable option for highly selected women who are truly refractory to all other treatments for detrusor overactivity incontinence. Such women should understand that Botox is not FDA-approved for this indication and that its use is experimental. No evidence identifies the optimal dose of Botox to be injected cystoscopically for incontinence related to detrusor overactivity. However, dosing information may be forthcoming from a Phase II study being performed by the company that markets Botox (Allergan).
Greatest risk is urinary retention
The most important immediate or short-term risk is probably urinary retention. Although experienced clinicians have estimated a low risk of retention, a much higher rate can be found when the postvoid residual is measured routinely after Botox injection. It still seems likely that Botox-associated retention is temporary, but it may last as long as the effect on symptoms, on the order of several months. Women should be counseled carefully to be sure they understand this.