Everything you need to know about the contraceptive implant
FDA approval means US women now have a progestin-only implant among their birth control options
IN THIS ARTICLE
Progestin implants have no abortifacient properties
Implanon is a safe choice for immediate postpartum contraception
Advise patients to use barrier contraception for 7 days after insertion
Bleeding irregularities are the most common reason for discontinuing the implant
On July 17, the US Food and Drug Administration (FDA) approved what may be the most effective hormonal contraceptive ever developed, a single-rod implant that goes by the trade name Implanon. The implant contains 68 mg of etonogestrel (ENG), the active metabolite of desogestrel, in a membrane of ethylene vinyl acetate. In clinical trials involving 20,648 cycles of exposure, only 6 pregnancies occurred, for a cumulative Pearl Index of 0.38 per 100 woman-years.1
This article reviews:
- the 2 contraceptive mechanisms
- indications and patient selection
- pharmacology, safety, adverse effects
- patient satisfaction and discontinuation rates
- insertion and removal
- key points of patient counseling
Unlike Norplant, a multi-rod implant which garnered a million American users before it was removed from the market, the single-rod implant is easy to insert and remove. Before you can order the implant, you must complete a manufacturer-sponsored training program.
Which patients are suitable candidates?
Because the subdermal implant contains only progestin, provides up to 3 years of protection, and requires no daily, weekly, or even monthly action on the part of the user, it is well-suited for:
- Women who wish to or need to avoid estrogen
- Teens who find adherence to a contraceptive regimen difficult
- Healthy adult women who desire long-term protection
- Women who are breastfeeding
Pros and cons
Advantages include:
- Cost. A study2 of 15 contraceptive methods found the implant cost-effective compared to short-acting methods, provided it was used long-term. As of press time, the manufacturer had not released the price.
- Short fertility-recovery time.
- No serious cardiovascular effects.3
Drawbacks. Progestin-only contraceptives also have disadvantages:
- Implants require a minor surgical procedure by trained clinicians for insertion and removal.
- Cost-effectiveness depends on duration of use; early discontinuation negates this benefit.2
- The implant does not protect against sexually transmitted infections—this is a disadvantage of all contraceptive methods except condoms and, perhaps, other barrier methods.
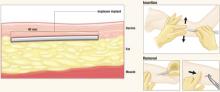
After Implanon is inserted just below the dermis 6 to 8 cm above the elbow crease on the inner aspect of the arm, it remains effective for up to 3 years. To remove it, make a 2-mm incision at the distal tip of the implant and push on the other end of the rod until it pops out.
The implant resides beneath the dermis but above the subcutaneous fat. It remains palpable but invisible, releasing about 60 μg of etonogestrel per day
Images: Rich Larocco
The Norplant experience
Research and development of progestin-only subdermal implants began more than 35 years ago, but early research involving very-low-dose implants found that they did not prevent ectopic pregnancies. This problem ended with Norplant, a 6-capsule implant using the potent progestin levonorgestrel (LNG).
Norplant was highly effective. Over 7 years of use, fewer than 1% of women became pregnant.4 Despite low pregnancy rates and few serious side effects, limitations in component supplies and negative media coverage on complications with removal led to its withdrawal in 2002, leaving no implant available in the US.5
The antiestrogenic effect
Long-acting, progestin-only contraceptives such as the new implant, the LNG-releasing intrauterine system (Mirena), and injectable methods (Depo-Provera) are safer than oral contraceptives (OCs) because they lack estrogen, which can provoke deep venous thrombosis.6,7
LNG, the gonane progestin used in Norplant, binds with high affinity to the progesterone, androgen, mineralocorticoid, and glucocorticoid receptors, but not to estrogen receptors. ENG, also known as 3-keto-desogestrel, demonstrates no estrogenic, anti-inflammatory, or mineralocorticoid activity, but has shown weak androgenic and anabolic activity, as well as strong antiestrogenic activity.
Unlike LNG, which binds mainly to sex hormone-binding globulin, ENG binds mainly to albumin, which is not affected by varying endogenous or exogenous estradiol levels. The safety of ENG has been demonstrated in studies of combined estrogen-progestin OCs and progestin-only OCs that use desogestrel as a component.
The progestin-only implant has 2 primary mechanisms:
- Inhibition of ovulation
- Restriction of sperm penetration of cervical mucus28
Ovulation is suppressed. LNG implants disrupt follicular growth and inhibit ovulation by exerting negative feedback on the hypothalamic–pituitary axis, causing a variety of changes, from anovulation to insufficient luteal function. A few women using LNG implants have quiescent ovaries, but most begin to ovulate as LNG blood concentrations gradually fall.29 The ENG implant suppresses ovulation by altering the hypothalamic–pituitary–ovarian axis and down-regulating the luteinizing hormone surge, which is required to support the production, growth, and maturation of ovarian follicles.11
Oocytes are not fertilized, even if follicles grow during use of the progestin implant. If the follicle ruptures, abnormalities of the ovulatory process prevent release of a viable egg.
Sperm cannot penetrate the cervical mucus. The antiestrogenic action of the progestin renders the cervical mucus viscous, scanty, and impenetrable by sperm.12
Contraceptive effects occur before fertilization
No signs of embryonic development have been found among implant users, indicating the implant lacks abortifacient properties.