New choices in prenatal screening for Down syndrome
A FASTER trial investigator discusses first-trimester combined screening, integrated screening, and findings from the trial
Imaging expertise is key
While it is beyond the scope of this article to detail the methodology of nuchal translucency measurement, specialized training and ongoing quality assurance are necessary to get the measurement right. Both the Society for Maternal-Fetal Medicine in the United States and the Fetal Medicine Foundation in the United Kingdom provide training and credentialing in nuchal translucency sonography. If performed correctly, it is an excellent screening marker. However, if attempted with no hands-on training, this imaging method yields unreliable results.
FIGURE 1 Nuchal translucency values increase with gestational age
Nuchal translucency values, in millimeters, in 561 pregnancies between 10 and 13 completed weeks of gestation, as estimated by crown-rump length. Reprinted with permission from Drs. Wald and Schucter; data from Schuchter K, et al.9
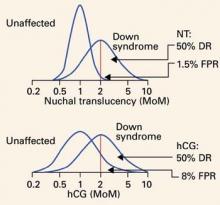
FIGURE 2 Very few unaffected fetuses have elevated nuchal translucency values
Distribution of values of second-trimester human chorionic gonadotropin (hCG) and first-trimester nuchal translucency among unaffected and Down syndrome pregnancies, both given in multiples of the median (MoM). Note that the unaffected nuchal translucency distribution is much narrower and taller than the unaffected hCG distribution. The scale is a log progression of increasing MoM values because both markers are log-normally distributed in unaffected and Down syndrome gestations. DR=detection rate; FPR=false-positive rate; NT=nuchal translucency.
2 important studies
The SURUSS trial, conducted mainly in the United Kingdom, was an observational study in which all women underwent first-trimester ultrasound measurement of nuchal translucency, as well as first- and second-trimester blood and urine sampling, with all samples stored. After all outcomes were chronicled, case-control sets of first- and second-trimester samples were constructed and assayed for a wide variety of known and potential screening markers. Nuchal translucency data also were analyzed. More than 48,000 women were enrolled, and 101 Down syndrome pregnancies were identified and studied. 2,10
The FASTER trial, completed more recently, was an observational study conducted at 15 enrollment centers in the US and involving more than 38,000 women, among whom 117 Down syndrome cases were identified.1 All women in the trial underwent first-trimester ultrasound examination and blood sampling between 10 weeks 3 days, and 13 weeks 6 days, and were asked to return for a second-trimester blood draw between 15 and 18 completed weeks, after which a report was issued detailing the separate results. In addition, combinations of markers across the trimesters were modeled and compared.
Perhaps the most interesting and surprising finding from the 2 studies is the remarkable similarity of results (TABLE). For 2 large populations on separate continents with a different ethnic mix, the primary findings were almost identical:
- 86% to 87% detection rate at a 5% false-positive rate for first-trimester combined screening (nuchal translucency, PAPP-A, and free β-hCG) and 80% to 83% detection rate at a 5% false-positive rate for second-trimester quad screening (AFP, uE3 , hCG, and inhibin A). In both trials, first-trimester screening was incrementally better than second-trimester screening, but the difference was not statistically significant.
- For integrated screening, a detection rate of 86% to 88% at a false-positive rate of 1%. Nuchal translucency and PAPP-A were measured during the first trimester, and quad markers during the second trimester. At a 5% false-positive rate, the detection rate in both trials was about 95%. With a serum-only integrated test, detection rates were 87% to 88% with a 5% false-positive rate.
- Nuchal translucency was more informative than any of the serum markers tested. This corroborates the rich literature on nuchal translucency published over the past decade. Both trials demonstrated that nuchal translucency measurement is effective when training is adequate, and that ongoing monitoring of quality is essential. However, in both studies, a satisfactory nuchal translucency measurement was not attained in about 7% of all women scanned between 10 and 13 weeks, so a small but significant number of women will not have nuchal translucency included in their risk assessment.
Other notable FASTER findings
Other findings from the FASTER trial that merit special attention:
- An ultrasound finding of cystic hygroma warrants an immediate prenatal diagnostic workup and was associated with an aneuploidy rate of 50% (one third of which was Down syndrome) and adverse outcomes in the great majority of cases.11 Cystic hygroma is uncommon, occurring in about 1 in 300 pregnancies in the first trimester.
- The value of the fetal nasal bone as a first-trimester ultrasound marker is unclear. In the FASTER trial, the nasal bone was studied in about 6,000 of the 38,000 ultrasound examinations, with no detected benefit. Thus, in a nonselected pregnant population, the nasal bone may not be a reasonable marker.12
- Various first- and second-trimester markers are modestly informative about adverse pregnancy outcomes other than aneuploidy (eg, fetal growth restriction, early delivery, and preeclampsia). Only a small proportion of affected pregnancies will be identified by these markers, alone or in combination.13,14