Simplified Foley insertion for cervical ripening
When a urologic sound is used as a stiffener, the clumsy, uncomfortable process of Foley-catheter positioning is transformed into a straightforward tactile insertion. The author details this simple, effective technique and discusses its advantages over alternative methods.
- A 16-Foley catheter allows the stiffener to pass through with ease; a 30-mL balloon allows for maximum balloon circumference utilizing a minimal amount of fluid.
- Hold the Foley catheter with internal stylet between the first 2 fingers of your dominant hand, then insert the ripener into the patient’s vagina up to the cervix.
- Position a finger on either side of the cervical opening, slide the catheter into the os until it touches the fetal vertex, and inflate the balloon.
- If the Foley is not spontaneously expelled, deflate the balloon and remove catheter within 5 to 12 hours, depending on time of induction.
- The Foley balloon is inexpensive and safe to use after ruptured membranes or in a trial of labor following a previous cesarean.
Postterm pregnancy, hypertensive disorders, diabetes mellitus, premature rupture of membranes, chorioamnionitis, perceived intrauterine growth restriction or macrosomia, oligohydramnios—these are just a few of many conditions that may call for induction of labor via cervical ripening. Historically, though, this process has been laborious due to the difficult insertion of mechanical agents and the adverse effects of pharmacologic therapies.
When a urologic sound is added as a stiffener to typical Foley balloon catheter insertion, however, cervical ripening becomes a much simpler, straightforward procedure. Here I will describe proper placement techniques and detail this procedure’s benefits over other, more arduous methods.
A brief history
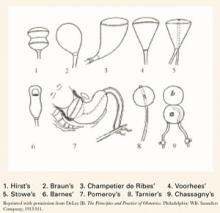
For many years, only mechanical means of ripening were available. In 1851, the intravaginal colpeurynter was used, followed by various intrauterine metreurynters and pear-shaped inflatable rubber bags inserted through the cervix extra-amniotically. Prolonged traction also was used to effect effacement and dilatation. (FIGURE 1)1,2 Later, natural or artificial laminaria or dilataria were implemented.3 But these mechanical methods fell into disfavor due to placement difficulty and concern about infection. Pharmacologic techniques, including vaginal insertion of gels, suppositories, or off-label oral agents, supplanted these earlier methods. However, the complications and side effects—including hyperstimulation (tachysystole, fetal distress, uterine rupture), nausea and vomiting, diarrhea, and fever—were troublesome.
Foley catheter. More than 25 years ago, the Hospital of the University of Pennsylvania instructed its residents in the standard Foleycatheter–insertion ripening technique. During our residencies there, my colleagues and I found this technique to be “cumbersome, somewhat archaic, and aesthetically suboptimal.”4 In 1995, recognizing the need for a safe, simple, effective, inexpensive, and easily reversible method of ripening with minimal side effects, I revisited the Foley balloon catheter—this time using a urologic sound or stiffener to ease insertion. This coupling transformed the awkward visual positioning of the Foley catheter through the endocervical canal into a straightforward tactile placement, similar in ease of accomplishment to the attachment of a scalp electrode to the fetal vertex.
Several reports in the English-language literature on the specifics of Foley ripening have discussed the use of various catheter sizes (14 Foley to 26 Foley) and balloon capacities (25 mL to 50 mL).5-37 In my practice, I use a 16-Foley catheter, which is large enough for the urologic sound to pass through with ease, and a 30-mL balloon, since a larger balloon may burst in the extra-amniotic space.1 Further, 30 mL—three 10-mL vials—of physiologic saline allows for maximum expansion of balloon circumference utilizing a minimal amount of fluid (TABLE 1). When balloons are inflated beyond 30 mL, expansion is linear along the catheter tubing, forming a cylindrical rather than spherical shape. Increasing the amount of fluid, therefore, provides no increased pressure on the cervix, but rather, displaces the presenting part.
A literature review reveals that investigators for 2 reports utilized nondescript Foley stiffeners or introducers, but ultimately dismissed them as unnecessary. In fact, the researchers concluded that the Foley stiffener could cause membranes to rupture prematurely.31,32 Since 1995, however, this technique has successfully been utilized in at least 113 patients at our institution. With the use of a blunt-tip stylet, we have not seen any rupture of membranes.
FIGURE 1 Various balloon dilators
TABLE 1
Comparison of instilled PSS (in mL) with Foley balloon circumference (in mm)
| PSS | 0 | 10 | 20 | 30 | 40 | 50 | 60 | 70 | 80 | 90 |
| CIRCUMFERENCE | 22 | 61 | 99 | 114 | 122 | 131 | 133 | 138 | 141 | 145 |
| PSS=physiologic saline solution | ||||||||||
Technique
Patient preparation. First, counsel the patient and obtain her informed consent. Prior to examination, ensure that a sonogram reading does not show signs of placenta previa; there are no visible lesions or a history of herpes; and gonorrhea, chlamydia, and group B streptococcus (GBS) cultures are negative. If the GBS culture is positive, provide appropriate intravenous antibiotic therapy during the ripening and later induction processes. Routinely examine the patient to elicit a Bishop score and confirm fetal vertex presentation.3 Secure a reactive nonstress test prior to Foley insertion.