2016 Update on cervical disease
The future of treatment for cervical cancer involves therapeutic vaccines and T-cell therapy. What you should know. Plus, follow-up data on HPV primary screening.
In this article
• The success of adoptive T-cell therapy
• Long-term follow-up of primary HPV screening
For the past 40 to 50 years, the first-line treatment for high-grade cervical intraepithelial neoplasia (CIN) has been excisional procedures (including loop electrosurgical excision [LEEP], cone biopsy, cryosurgery, and laser therapy), and these treatments work well. It appears, however, that these procedures potentially can lead to preterm birth.1–3 With results from large, comprehensive meta-analyses that control for such risk factors as smoking and other factors that could contribute to both preterm birth and high-grade CIN, we have learned that excision treatment can result in a 2% to 5% increased risk for preterm birth, depending on the size and the extent of excision performed.1–3 The preterm birth rate in the United States is about 11.4%.4 With about 500,000 excisional treatments for high-grade CIN performed in the United States every year, and about 2% of preterm births caused by excisional procedures, conservatively, about 5,000 to 10,000 US preterm births are directly related to excisional procedures for high-grade CIN annually.
Clearly, excisional treatment for high-grade CIN and its connection to preterm birth adds to health care costs and long-term morbidity because babies that are born preterm potentially have diminished functionality. We need a better treatment approach other than excision to CIN, which is known to be a virally mediated disease. Consider the fact that just because excisional procedures remove potentially cancerous cells does not mean that these treatments remove the underlying reason behind the high-grade CIN—HPV. We cannot cut out a virus. Consequently, many studies have explored better-targeted therapies against high-grade CIN. Immune-based therapies, which can train a patient’s own immune system to attack HPV-infected cells, are exciting possibilities.
In this Update, I focus on 2 studies of immune-based therapies to treat cervical cancer. In addition, I discuss long-term follow-up data that are available regarding efficacy of primary HPV testing.
HPV therapeutic vaccine shows promise in RCT
Trimble CL, Morrow MP, Kraynyak KA, et al. Safety, efficacy, and immunogenicity of VGX-3100, a therapeutic synthetic DNA vaccine targeting human papillomavirus 16 and 18 E6 and E7 proteins for cervical intraepithelial neoplasia 2/3: a randomised, double-blind, placebo-controlled phase 2b trial. Lancet. 2015;386(10008):2078-2088.
While the promise of immune-based therapies to target a virally mediated disease has good scientific rationale, there have been many generally negative studies published in the past 15 years on immune-based targeted therapies. This study by Trimble and colleagues has interesting results because it is a randomized controlled trial (RCT) using a DNA vaccine delivered with a novel approach called electroporation. Electroporation generates a small electrical shot at the vaccine site that potentially increases a vaccine's DNA uptake and the patient's immune response.
Details of the study
Women aged 18 to 55 years with HPV16- or HPV18-positive high-grade CIN from 36 academic and private gynecology practices in 7 countries were assigned in a 3:1 blinded randomization to receive vaccine (6 mg; VGX-3100) or placebo (1 mL), given intramuscularly at 0, 4, and 12 weeks. Patients were stratified by age 25 or older versus younger than 25 and by CIN2 versus CIN3. The primary efficacy endpoint was regression to CIN1 or normal pathology 36 weeks after the first vaccine dose.
A mandatory interim safety colposcopy was performed 12 weeks after the third vaccine dose. At 36 weeks (the primary endpoint visit), patients with colposcopic evidence of residual disease underwent standard excision (LEEP or cone). In patients with no evidence of disease, investigators could biopsy the site of the original lesions. At 40 weeks, when all patients had completed their first visit after the primary endpoint, the data were unmasked. Long-term follow-up data were collected on all patients with remaining visits. Patients and study site investigators and personnel stayed masked to treatment until study data were final.
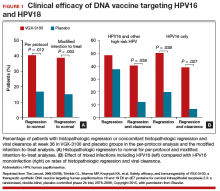
Results indicated a significant clinical response as well as an immune response in those patients who were treated with electroporation and the vaccine versus electroporation and placebo. In the per-protocol analysis, 53 (49.5%) of 107 vaccine recipients and 11 (30.6%) of 36 placebo recipients had histopathologic regression (percentage point difference [PPD], 19.0 [95% confidence interval CI, 1.4-36.6]; P = .034) (FIGURE 1). In the modified intention-to-treat analysis, 55 (48.2%) of 114 vaccine recipients and 12 (30.0%) of 40 placebo recipients had histopathologic regression (PPD, 18.2; 95% CI, 1.3-34.4; P = .034).
Injection-site reactions occurred in most patients, but only erythema was significantly more common in the vaccine group than in the placebo group (PPD, 21.3 [95% CI, 5.3-37.8]; P = .007).
What this evidence means for practice
In prior studies of immunotherapies, there have not been good correlations between immune responses and clinical responses, and this is one of the important differences between this study by Trimble and colleagues and prior studies in this space. Unfortunately, immune-based therapies are a "shot in the dark," with researchers not knowing which patients may have an increased immune response but no clinical response or a clinical response but no immune response. The measured immune responses are from peripheral blood, an immune response that might not reflect the milieu of immune responses in the cervical-vaginal tract.
If perfected, technologies like these hold the promise of minimizing the amount of patients who need to undergo excisional procedures because patients' own immune systems have been trained to target HPV-infected cells. The bigger hope is that we will be able to minimize preterm births that are directly related to treatment of dysplasia.