Warfarin therapy: Tips and tools for better control
Monitoring patients on warfarin therapy is challenging. The tools highlighted here—from online forums and Web-based dosing calculators to patient education materials and self-monitors—can help.
• INR testing by an anticoagulation management service or private clinician can be reduced to intervals of as long as 4 weeks, but should be more frequent when dosing adjustments occur. B
• Weekly patient self-testing is associated with comparable clinical outcomes to high-quality clinic-based anticoagulation management. A
• Patients who self-test (and report their results) weekly should test more frequently when a change in medication (including herbal remedies and dietary supplements) or diet or an illness occurs. C
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Approximately 4 million Americans are receiving longterm oral anticoagulation therapy to reduce the risk of primary and secondary thromboembolism.1,2 And, as the population ages, the number of patients on lifelong therapy with warfarin—the only oral anticoagulant available in the United States until dabigatran was approved by the US Food and Drug Administration late last year3—is expected to grow.4
Such patients present a challenge for family physicians. Warfarin is notorious for having both a narrow therapeutic index and numerous drug and dietary interactions.5,6 To safeguard patients on warfarin therapy, frequent, and diligent, monitoring is required.
Engaging patients as participants in their own care can help you decrease the hazards. With that in mind, this article features warfarin treatment tips and tools for both physicians and patients, along with a review of some basic safeguards.
Warfarin therapy: Keeping it safe
Warfarin, a vitamin K antagonist, is used to prevent systemic embolism in patients with prosthetic heart valves, atrial fibrillation, or inherited/acquired thrombophilic disorders; as an adjunct in the prophylaxis of systemic embolism after myocardial infarction (MI); and to reduce the risk of recurrent MI, as well as venous thromboembolism.4,7 Because there is a small but definite risk (1%-2% per year)8 of severe bleeding associated with warfarin, however, therapy should be initiated only when the potential benefits clearly outweigh the risks.
A major contraindication for warfarin therapy is early pregnancy. The anticoagulant is a teratogen, causing deformations of the face (depressed nasal bridge) and bones (stippled epiphyses), neonatal seizures, and spontaneous abortion. If a woman in the first trimester of pregnancy requires anticoagulation, low-molecular-weight heparin should be substituted instead.9
In fact, warfarin is not recommended in the second or third trimesters either, as the use of vitamin K antagonists increases the risk of miscarriages, structural defects, and other adverse outcomes. Nor is warfarin recommended for women who are planning to become pregnant.
Warfarin is also contraindicated in patients for whom the risk of major bleeding outweighs the benefits. Risk factors for warfarin-associated bleeding include renal insufficiency and concomitant antiplatelet therapy, and physicians can use published clinical prediction rules to estimate bleeding risk.10
Dosing considerations
When you start a patient on warfarin therapy, it is important to ensure that therapeutic concentrations are achieved in a timely manner—and that the risk of supra- and subtherapeutic international normalized ratio (INR) values—≥4.0 and <2.0, respectively—is minimized.6
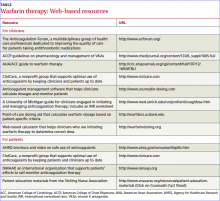
Factors to consider in determining the starting dose include patient-specifi c measures such as age, height, and weight; concomitant medications; and comorbidities. Increasing age, female sex, and a low body mass index all indicate a need for a lower dose.11 A number of Web-based dosing calculators (TABLE) can help clinicians estimate the therapeutic dose in patients who are new to warfarin.
Thyroid activity also affects warfarin dosing requirements.12 Hypothyroidism makes people less responsive to warfarin,13 while hyperthyroidism boosts the anticoagulant effect.14 Several mechanisms have been proposed for this effect, including changes in the rate of breakdown of clotting factors and in the metabolism of warfarin.15,16
Frequency of monitoring. Regardless of the initiation dose, INR values of outpatients should be monitored at least 2 to 3 times a week for the fi rst 7 to 10 days of therapy, or until a stable value is achieved. (In an inpatient setting, INR monitoring is usually performed daily until the therapeutic range has been maintained for ≥2 days.) The target INR level varies from case to case depending on the clinical indicators, but tends to be between 2 and 3 for most patients and between 2.5 and 3.5 for those with mechanical heart valves.17
After stabilization, testing can be reduced to intervals of as long as 4 weeks, althoughevidence suggests that more frequent testing leads to greater time-in-therapeutic range (TTR).18,19 When dosing adjustments are required, the cycle of more frequent monitoring should be repeated until a stable dose response can again be achieved.
Benefits of patient involvement
Patients on warfarin may be managed in one or more of the following 3 methods: (1) with usual care, provided by the patient’s personal physician; (2) by anticoagulation management services (AMSs), specialized programs overseen by physicians, pharmacists, and/or nurses; or (3) by self-testing/self-management, with the help of point-of-care devices that allow patients to monitor their own INR levels and adjust their anticoagulation dose, within certain limits, in consultation with a clinician.4