Postmenopausal osteoporosis: Another approach to management
The effectiveness of oral bisphosphonates is compromised by poor compliance. IV bisphosphonates provide another option.
Therapy underused after hip fracture. Most patients are not properly evaluated or treated for osteoporosis after a hip fracture. Zoledronic acid is the only bisphosphonate that has been specifically studied in this population to evaluate its effectiveness in preventing additional fractures. In the HORIZON-Recurrent Fracture Trial, patients were randomized to receive IV zoledronic acid 5 mg (n=1065) or placebo (n=1062) within 90 days after repair of a hip fracture and yearly thereafter.29 Compared with placebo, an annual infusion of IV zoledronic acid resulted in a 35% reduction in the rate of any new clinical fracture (13.9% vs 8.6%; P=.001; NNT to prevent 1 fracture at 3 years=19). BMD of the contralateral hip increased in the zoledronic acid group by 5.5% after 3 years compared with a 0.9% decrease in the placebo group (P<.001). In addition, there was a 28% reduction in all-cause mortality (9.6% for zoledronic acid vs 13.3% for placebo; P=.01).
This study demonstrated that a yearly infusion of zoledronic acid is effective in preventing subsequent clinical fractures in patients who have recently suffered a hip fracture and that it may reduce all-cause mortality. These results support the need for careful discharge planning after a hip fracture to include appropriate treatment to prevent subsequent fractures.
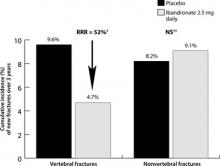
FIGURE 1
Daily oral ibandronate (2.5 mg*) reduced risk of vertebral fractures in the BONE study (n=982)
*Ibandronate 2.5 mg tablets are not available on the market.
†P<.001 vs placebo.
‡No significant difference vs placebo.
BONE, oral iBandronate Osteoporosis vertebral fracture trial in North America and Europe; RR, relative risk reduction.
Data from Chesnut CH II, et al. J Bone Miner Res. 2004.13
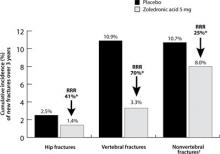
FIGURE 2
Intravenous zoledronic acid (5 mg, once yearly) reduced risk of fractures in the HORIZON-Pivotal Fracture Trial (n=7736)
*P≤.002 vs placebo.
†Hip fracture was not excluded from analysis of nonvertebral fracture.
HORIZON, Health Outcomes and Reduced Incidence with Zoledronic acid Once Yearly; RR, relative risk reduction.
Data from Black DM et al. N Engl J Med. 2007.28
GI issues and flu-like symptoms are among the side effects
Side effects of oral bisphosphonates include such GI problems as dyspepsia, nausea, and reflux. There is also a small risk of developing inflammation of the esophagus and gastric ulcers.
In some patients, IV bisphosphonates can cause transient flu-like symptoms (eg, myalgia, headache, pyrexia) within 3 days of administration.27-30 These symptoms are generally mild and last a few days, and they can be reduced by taking acetaminophen or ibuprofen for several days after the injection or infusion. Symptoms are uncommon with subsequent injections or infusions. Counsel patients about the possibility of these symptoms and how to manage them.
The safety of switching directly from weekly oral alendronate 70 mg to IV zoledronic acid 5 mg was evaluated in a 12-month randomized, double-blind study. The results showed that switching to zoledronic acid was well tolerated.31 The overall cost of IV annual zoledronic acid is similar to the annual cost of a branded oral bisphosphonate.
When to avoid bisphosphonates
Give no bisphosphonate, oral or IV, to a patient with significant renal impairment because the drugs are excreted renally. Be sure a patient’s calcium level is normal and that she is not vitamin D deficient. Due to the use of sunscreens and sun avoidance, vitamin D deficiency is common, even in sunny climates. Consider using vitamin D supplementation, as needed, before infusion of an IV bisphosphonate. And emphasize the importance of lifelong calcium and vitamin D supplementation to all patients with osteoporosis.
Osteonecrosis of the jaw (ONJ) in bisphosphonate-treated patients with osteoporosis is rare (<1 in 100,000 patient-years in non-cancer patients).32,33 Most cases of ONJ in this setting have occurred “in cancer patients treated with intravenous bisphosphonates undergoing dental procedures. Some cases have occurred in patients with postmenopausal osteoporosis treated with either oral or intravenous bisphosphonates.”34 A prescriber should perform a routine oral examination before initiating bisphosphonate treatment. Consider referring patients who have a history of concomitant risk factors (eg, cancer, chemotherapy, radiotherapy, corticosteroids, poor oral hygiene, pre-existing dental disease or infection, anemia, coagulopathy) for a dental examination and appropriate preventive dentistry.
While on treatment, patients with concomitant risk factors should avoid invasive dental procedures, if possible. If ONJ develops during bisphosphonate therapy, dental surgery may exacerbate the condition. If a dental procedure is unavoidable, no data are available to suggest whether discontinuation of bisphosphonate treatment reduces the risk of ONJ. The treating physician must rely on clinical judgment regarding the benefits/risks for an individual.34