A look at the long-term safety of an extended-regimen OC
This study reveals that adverse events after 4 years of continuous oral contraceptive use were similar to those observed during 1 year of use.
Statistical analysis
Descriptive statistics included the number of subjects, and the mean, median (where appropriate), standard deviation or standard error of the mean (SE), or minimum and maximum values of patient characteristics. We summarized discrete events using frequencies or percentages. As this study was designed primarily to be observational and to gain further long-term experience with the regimen, we did not conduct formal power analyses and sample size calculations. For contraceptive trials, the US Food and Drug Administration typically requires a minimum exposure of 200 women using the method for 1 year. We also omitted a formal efficacy analysis, as efficacy was established in the Phase 3 clinical program.1
Results
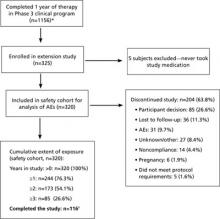
Of the 320 subjects enrolled and treated, 244 (76.3%) completed at least 1 year of treatment; 173 (54.1%) completed at least 2 years of treatment; and 85 (26.6%) completed 3 years of treatment in this extension study, beyond the 1 year completed in the Phase 3 clinical trials (FIGURE). A total of 204 women (63.8%) discontinued treatment; primarily due to personal decisions (26.6%), becoming lost to follow-up (11.3%), and adverse events (9.7%). These discontinuation rates are consistent with those in other long-term studies.6-8
FIGURE
Of the 320 participants enrolled, 116 completed the study
* In the pregnancy prevention study, 979 patients completed 1 year; in the endometrial safety study, 177 completed 1 year. Only 11 of the original 36 sites participated in the extension study, so not all 1156 subjects had the option of enrolling in the extension.
† Not all subjects enrolled at the same time. Thirty-one patients were participating in the study with various durations of exposure when the study was ended. Although they did not complete 3 full years of use, they did participate in the full course of the study that was available to them and were therefore classified as “completers.”
Serious adverse events were few
SAEs were reported by 12 subjects; 3 were possibly related to treatment—spontaneous abortion in a 33-year-old subject, nonthrombotic coronary artery spasm in a 40-year-old subject, and acute cholecystitis in a 37-year old subject. No venous thromboembolic events (VTEs) occurred; however, such events are rare (approximately 7-18 VTEs/100,000 OC users annually9) and would be unlikely in a study of 320 subjects.
Nonurgent adverse events comparable to earlier studies
The most commonly reported treatment-related AEs were headache (9.4%), metrorrhagia (9.1%), increased weight (6.9%), and dysmenorrhea (4.4%), as noted in TABLE 2. The most frequently reported treatment-emergent AEs (ie, regardless of relationship to study medication) were headache (21.9%), upper respiratory tract infection (18.4%), nasopharyngitis (15.0%), sinusitis (12.2%), and back pain (11.6%). A total of 31 subjects (9.7%) discontinued the study due to AEs. The incidence rates of treatment-emergent and treatment-related AEs in this study were not substantially higher than those in the Phase 3 trials.1
TABLE 2
Adverse events attributable to treatment occurred in ≥2% of participants (N=320)
| MedDRA System organ class and preferred term | n (%) |
|---|---|
| Reproductive system and breast disorders Metrorrhagia Dysmenorrhea | 29 (9.1) 14 (4.4) |
| Nervous system disorders Headache | 30 (9.4) |
| Investigations Weight increased | 22 (6.9) |
| Infections and infestations Vulvovaginal mycotic infection Vaginitis, bacterial Fungal infection | 13 (4.1) 9 (2.8) 7 (2.2) |
| Skin and subcutaneous tissue disorders Acne | 7 (2.2) |
| MedDRA, Medical Dictionary for Regulatory Activities. | |
Pregnancies due mostly to nonadherence
We conducted no formal efficacy analyses. Pregnancy was determined by a positive result on a pregnancy test conducted at the study site. Six subjects (1.9%) became pregnant during the study; 4 were noncompliant with the study medication, and 2 became pregnant at least 14 days after completing the study medication. One spontaneous abortion was reported. Among those participants who continued their pregnancies, none reported abnormal outcomes.
Laboratory values changed minimally, if at all
No notable changes occurred in serum chemistry, hematology, or urinalysis values. Specific mean changes from baseline included increases of 5.0 mg/dL for total cholesterol, 2.4 mg/dL for high-density lipoproteins, and 4.0 mg/dL for low-density lipoproteins; and decreases of 5.9 mg/dL for triglycerides and 0.1 g/dL for hemoglobin.
Vital signs remained stable
No notable changes occurred in systolic or diastolic blood pressure, heart rate, or temperature. The increase in mean weight that we observed (10.4 lb) is not unexpected, as the time period of evaluation was as long as 4 years after documentation of the baseline value.
Reported bleeding or spotting diminished over time
Median rates of unscheduled bleeding or spotting declined over the course of the study, from 4 days in 91 during cycle 1 to 1 day in 91 during cycle 11. In most of the 91-day cycles, participants consistently reported a median of 3 days of scheduled (withdrawal) bleeding or spotting.