Meningococcal vaccine for infants?

With the incidence of meningococcal disease declining in all age groups, ACIP is weighing the need for vaccination in children <2 years.
The Advisory Committee on Immunization Practices (ACIP) is evaluating whether to recommend the use of meningococcal vaccines for infants and children <2 years.1 The decision may be made within the next 4 to 8 months. In its deliberation, ACIP must consider several issues, which I review here.
Current and impending vaccine options. Two quadrivalent meningococcal conjugate vaccines (MCV4) are licensed by the US Food and Drug Administration (FDA) for use in the United States: Men ACWY-CRM (Menveo, Novartis) and Men ACWY-D (Menactra, Sanofi Pasteur).2 Both vaccines protect against 4 meningococcal serogroups (A, C, Y, and W-135) and are approved for use among those ages 2 to 55 years. In addition, Menactra was recently licensed as a 2-dose series for children ages 9 to 23 months. ACIP recommends routine use of MCV4 for adolescents ages 11 to 18 years, with a preference for the first dose at ages 11 to 12 years; and for all individuals between the ages of 2 and 55 years who are at increased risk for meningococcal disease ( TABLE ).
Complicating matters is the pending availability of more formulations. In addition to the 2-dose Menactra option for children 9 to 23 months, Novartis has an application before the FDA for a 4-dose schedule with Menveo, given at ages 2, 4, 6, and 12 months. GlaxoSmithKline has just received approval from the FDA for MenHibrix, a combination vaccine that contains antigens against Haemophilus influenzae type b (Hib) and 2 meningococcal serogroups, C and Y, licensed as a 4-dose series given at ages 2, 4, 6, and 12 months.
These vaccines have proven to be immunogenic in infants without diminishing the effectiveness of other, co-administered vaccines in normal infant populations. They also appear to be safe, although the studies to date have not been sufficiently large to detect uncommon adverse events.3-7
Table
Patients at high risk for meningococcal disease
|
| Source: CDC. Updated recommendations for use of meningococcal conjugate vaccines—Advisory Committee on Immunization Practices (ACIP), 2010. MMWR Morb Mortal Wkly Rep. 2011;60:72-76. |
Meningococcal disease incidence and prevalence are declining
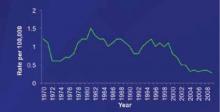
One major consideration for ACIP is the changing epidemiology of meningococcal disease and the low prevalence of disease in all age groups, including infants. The incidence of meningococcal disease has declined in the United States since 1980, with a marked and sustained decline since 2000 ( FIGURE 1 ).8 This decline has occurred in all age groups including infants, who have the highest rate of infection ( FIGURE 2 ).8 This decline in incidence occurred for all serogroups, including serogroup B.8
Serogroup B. Among children <5 years, including infants, half of meningococcal disease is caused by serogroup B,8 and these infections would not be prevented by any of the currently licensed vaccines or by those under review. Furthermore, half of all infections occur before age 9 months8 —an age range for which Menactra in not approved.
Serogroup C and Y. One-third of infections with serogroups C and Y occur before the age of 6 months8 and would not be prevented by any of the new products. Also of note: From 2007 to 2009, the mean number of cases of serotype A or C infection occurring each year in children <5 years was 77.8
The impact on children vs adults. Meningococcal disease in children is generally less severe than that occurring in older age groups. Overall case fatality in children is 6%; 10% in those with serogroup B and 1% in those with serogroup Y.8 The disease in children does result in significant sequelae, however, with 10% suffering hearing loss and 1% to 2% requiring amputation. From 2007 to 2009, there were 4 to 8 deaths per year among children under age 5, and 8 to 12 children per year experienced serious sequelae.8
FIGURE 1
The incidence of meningococcal disease has declined steadily since 20008
1970-1996 National Notifiable Diseases Surveillance System data. 1997-2009 Active Bacterial Core surveillance data estimated to the US population.
FIGURE 2
Meningococcal disease has declined among infants and other age groups8
Active Bacterial Core surveillance cases from 1993-2009 estimated to the US population with 18% correction for underreporting.
ACIP’s dilemma
The low morbidity and mortality associated with meningococcal disease is one issue to consider when deciding whether to recommend new vaccines as part of the routine infant and child immunization schedule. The vaccine schedule is already crowded and complex, and parents increasingly are questioning the need for additional antigens.