MODULE 1: Historical Review of Evidence-Based Treatment of Hypertension
MODULE 1: Historical Review of Evidence-Based Treatment of Hypertension
MODULE 3: Using Thiazide-Type Diuretics in African Americans with Hypertension
,Dr White is President of the American Society of Hypertension (voluntary unpaid position) and is a paid cardiovascular safety consultant to Abbott Immunology; Ardea Biosciences, Inc; AstraZeneca; BioSante Pharmaceuticals, Inc; Forest Research Institute, Inc; Novartis Corporation; Roche Laboratories, Inc; and Takeda Global Research & Development Center, Inc. He holds no stocks in pharmaceutical companies and is not a member of any speakers’ bureaus.
Introduction
Hypertension is a transformative condition in modern medicine due to the various numeric definitions of the disease, the decision of when to initiate therapy and to what level to treat, and the evolution of our understanding of the long-term complications of the hypertensive disease process. Hypertension is notable as 1 of the first conditions that only rarely manifested symptoms and whose eventual sequelae could take years, if not decades, to become known. In addition, hypertension was the first condition in which clinicians initiated therapy for patients who were otherwise healthy. Hypertension also led to 1 of the first screening programs for any disease as well as the first robust preventive effort for a chronic medical condition.1
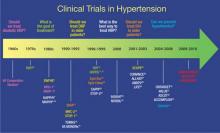
Clinical trials have been performed for 5 decades to evaluate the potential benefits of lowering blood pressure (BP) in patients with hypertension and its comorbid conditions (FIGURE 1).2-37 The first clinical trial to identify the increased risk of cardiovascular (CV) mortality related to hypertension was published in the mid-1960s.2,38 In fact, the Veterans Administration (VA) Cooperative trial was the first randomized, placebo-controlled, double-blind, multi-institutional drug efficacy trial ever conducted in CV medicine.38 It involved 143 men who met the 1964 definition of hypertension (ie, diastolic BP [DBP] ≥115 mm Hg) and who were randomized to either triple therapy with low doses of hydrochlorothiazide (HCTZ), reserpine, and hydralazine, or to placebo. The trial was terminated early when, after 18 months of treatment, rates of morbidity and mortality were substantially lower in the treated group than in the placebo group.2 The trial was the first to confirm that antihypertensive treatment, even in patients with existing CV damage and significant hypertension, could dramatically reduce the incidence of stroke, congestive heart failure (CHF), and progressive kidney damage.38
FIGURE 1
Clinical trials in hypertension during the past 5 decades
ACCOMPLISH, Avoiding Cardiovascular Events Through Combination Therapy in Patients Living With Systolic Hypertension trial; ALLHAT, Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial; ALTITUDE, Aliskiren Trial in Type 2 Diabetes Using Cardiovascular and Renal Disease Endpoints; ANBP1, Australian National Blood Pressure Study 1; ANBP2, Australian National Blood Pressure Study 2; ASCOT, Anglo-Scandinavian Cardiac Outcomes Trial; ATMOSPHERE, Efficacy and Safety of Aliskiren and Aliskiren/Enalapril Combination on Morbi-mortality in Patients With Chronic Heart Failure study; CAPPP, Captopril Prevention Project; CONVINCE, Controlled Onset Verapamil Investigation of Cardiovascular End Points trial; DBP, diastolic blood pressure; EWPHE, European Working Party on High Blood Pressure in the Elderly study; HAPPHY, Heart Attack Primary Prevention in Hypertension trial; HBP, high blood pressure; HDFP, Hypertension Detection and Follow-up Program; HOT, Hypertension Optimal Treatment study; INSIGHT, International Nifedipine GITS Study of Intervention as a Goal in Hypertension Treatment; I-PRESERVE, Irbesartan in Heart Failure with Preserved Systolic Function study; ISH, isolated systolic hypertension; LIFE, Losartan Intervention For Endpoint Reduction in Hypertension trial; MAPHY, Metoprolol Atherosclerosis Prevention in Hypertensives study; MRC-1, Medical Research Council trial of treatment of mild hypertension; MRC-2, Medical Research Council trial of treatment of hypertension in older adults; NORDIL, Nordic Diltiazem study; ONTARGET, Ongoing Telmisartan Alone and in Combination with Ramipril Global Endpoint Trial; SCOPE, Study on Cognition and Prognosis in the Elderly; SHEP, Systolic Hypertension in the Elderly Program; STOP-1, Swedish Trial in Old Patients with Hypertension-1; STOP-2, Swedish Trial in Old Patients with Hypertension-2; Syst-China, Systolic Hypertension in China trial; Syst-Eur, Systolic Hypertension in Europe trial; TOMHS, Treatment of Mild Hypertension Study; TROPHY, Trial of Preventing Hypertension; UKPDS, United Kingdom Prospective Diabetes Study; VA, Veterans Administration; VA MONORx, VA Monotherapy of Hypertension study; VALUE, Valsartan Antihypertensive Long-term Use Evaluation trial.Although the Framingham Study (www.framinghamheartstudy.org) was, of course, one of the seminal studies in the field of CV medicine, it was observational in nature, rather than interventional like most of the studies highlighted in this article.
The Medical Research Council trial of the treatment of mild hypertension (MRC-1) (ie, defined as DBP 90-109 mm Hg) demonstrated that a significant reduction in DBP among individuals receiving the diuretic bendroflumethiazide or the beta-blocker propranolol significantly reduced the rate of stroke compared with placebo, with a rate of 1.4 per 1000 patient-years of observation in the treatment group vs 2.6 per 1000 patient-years in the placebo group (P < .01).5 The treatment group also had significantly lower rates of all CV events than the placebo group, and this difference was statistically significant (P < .05). However, the treatment groups experienced significantly increased rates of adverse effects compared with placebo.5