Androgenetic Alopecia: What Works?
Second, long-term data are nonexistent for most of our AGA treatments. The exceptions include finasteride, dutasteride, and topical minoxidil, which have reasonably adequate long-term studies.4-6 However, most other treatments have been evaluated only through short-term studies. It is tempting to assume that results from a 24-week study can be used to infer how a patient might respond when using the same treatment over the course of many decades; however, making these assumptions would be unwise.
Third, most AGA treatments help improve hair density in only a proportion of patients who decide to use the given treatment. There usually is one subgroup of patients for whom the treatment does not seem to help much at all and one subgroup for whom the treatment halts further hair loss but does not regrow hair. For example, in the case of finasteride treatment of male AGA, approximately 10% of patients do not seem to respond to treatment at all, and another 50% seem to be able to halt further loss but never achieve hair regrowth.7 In an analysis of 12 studies with 3927 male patients, Mella et al8 showed that 5.6 patients needed to be treated short term and 3.4 patients needed to be treated long term for 1 patient to perceive an improvement in the hair. It is clear that many males who use finasteride will not see evidence of hair regrowth. This same general concept applies for all available treatments and is important to remember if a patient with AGA decides to start 2 new treatments simultaneously. Consider the 34-year-old man who starts oral minoxidil and platelet-rich plasma (PRP) for AGA. At his follow-up appointment 9 months later, the patient reports improved hair density and wants to know what contributed to the improvement: the oral minoxidil, the PRP, or both? Many practitioners would believe that both treatments likely provided some degree of benefit—but in reality, that represents a flaw in logic. If 2 hair loss treatments are started at exactly the same time, it is impossible to know the relative benefit of each treatment and whether one might not be helping at all. Combination therapies are still common in my practice and highly encouraged, but my personal preference is to stagger start dates whenever possible so I can determine each treatment’s contribution to the patient’s final outcome.
Finally, when evaluating what works for AGA, we need to define the specific patient subpopulation, as the available data are less robust for some patient groups than others. We have limited data in children and adolescents with AGA, as well as limited comparative data across different racial backgrounds, body mass indices, and underlying health issues. For example, data on the most effective strategies to treat female AGA in the setting of polycystic ovary syndrome, premature menopause, and other endocrine disorders are lacking.
Which Treatments Also Have Good Safety?—The treatment that a patient ultimately selects also depends on its actual or perceived safety. Patients have vastly different levels of risk tolerance. Some patients would much rather start a less effective treatment if they believe that the chances of experiencing treatment-related adverse effects would be lower. In general, topical and injectable treatments tend to have fewer adverse effects than oral therapies. Long-term safety data generally are lacking for many hair-loss therapies. A limited number of studies of topical minoxidil include data up to 5 years,4 and some studies of oral finasteride and oral dutasteride include patients who used these medications for up to 10 years.5,6
So Then, What Works?
The Table shows treatments for AGA and how I prioritize starting them in my own clinic. First-line treatment options often include those with level 1 evidence but also may include those with less-robust evidence plus a good history (over many years) of safety, affordability, ease of use, and effectiveness (eg, spironolactone and finasteride for female-pattern hair loss).
• Male AGA: I consider topical minoxidil, oral finasteride, and oral dutasteride as first-line agents, and low-level laser, PRP, oral minoxidil, and topical finasteride as second-line agents. Only topical minoxidil and oral finasteride are approved by the US Food and Drug Administration (FDA) for AGA in males; laser devices are FDA cleared.
• Premenopausal females with AGA: I use topical minoxidil and spironolactone as first-line agents. Low-level laser, PRP, oral minoxidil, and oral contraceptives are helpful second-line agents. Only topical minoxidil is FDA approved in women. I consider all treatments, with the exception of low-level laser, to be contraindicated in pregnancy.
• Postmenopausal females with AGA: I consider topical minoxidil, spironolactone, and oral finasteride as first-line agents. Low-level laser, PRP, oral minoxidil, and oral dutasteride are helpful second-line agents.
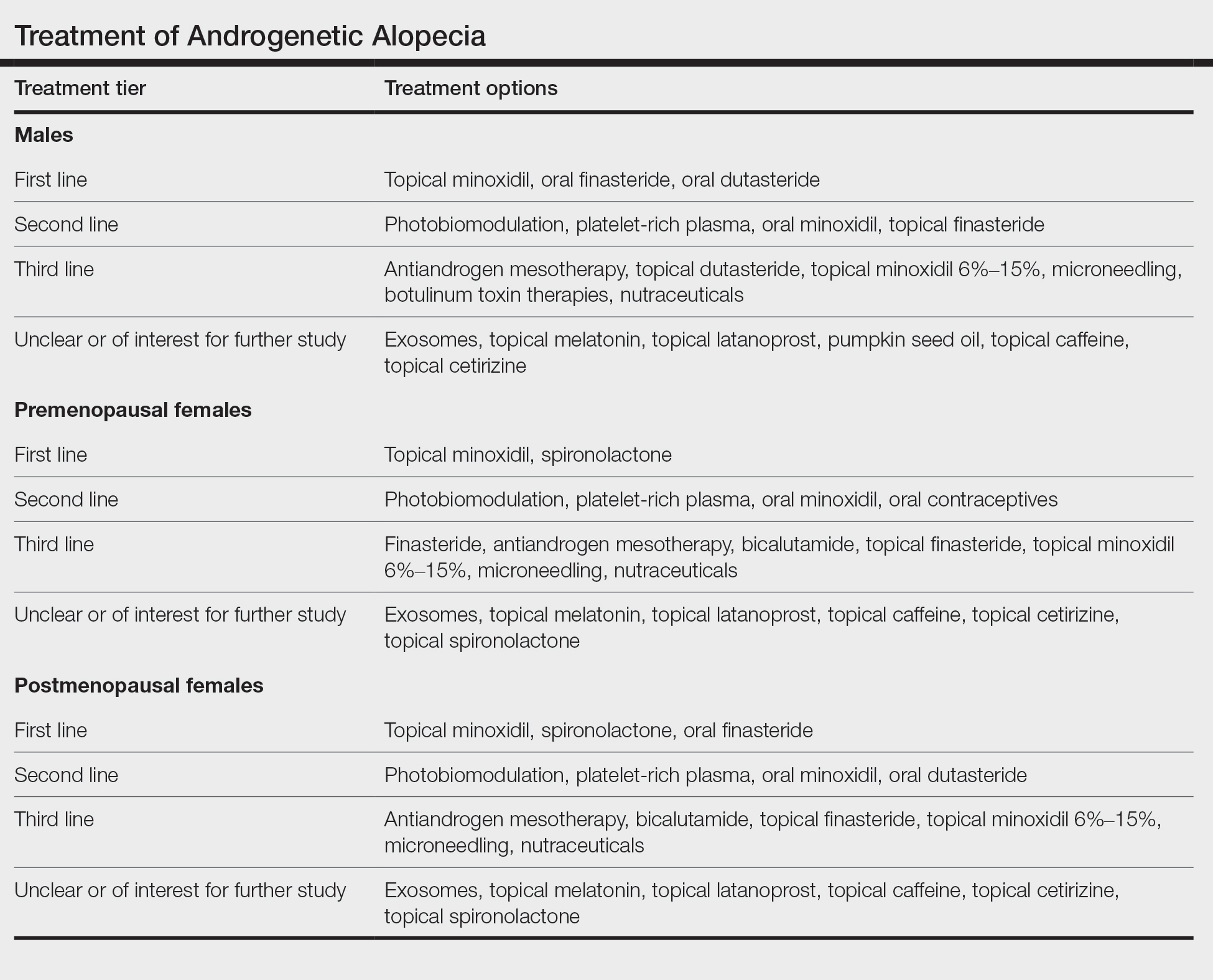
When choosing an initial treatment plan, I generally will start with one or more first-line options. I will then add or replace with remaining first-line options or a second-line option after 6 to 12 months depending on how well the patient responds to the first-line options. Patients who do not wish to use first-line options or have contraindications begin with second-line options. Third-line options are best reserved for patients who do not respond to or do not wish to use first- and second-line options.
Experts differ in opinion as to what constitutes a first-line treatment option and what constitutes a second- or third-line option. For example, some increasingly consider oral minoxidil to be a first-line option for AGA.9 In my opinion, the lack of high-quality comparative, randomized, controlled trials and long-term safety data keep oral minoxidil reserved as a respectable second-line option. Similarly, some experts reserve oral dutasteride as a second-line option for AGA.10 In my opinion, the data now are of the highest-quality evidence (level 1)9 to support placing oral dutasteride in the tier of first-line treatments.
Shared decision-making using an evidence-based approach is ultimately what connects patients with treatment plans that offer a good chance of helping to improve hair loss.






