An Analysis of the Clinical Trial Landscape for Cutaneous Melanoma
Recognizing current trends and obstacles in melanoma clinical trial development is critical to future progress in this field. This article examines the melanoma research enterprise to identify changing trends and potential barriers to success. All trials registered at ClinicalTrials.gov after September 2005 were evaluated for possible inclusion. A total of 777 interventional trials designed specifically for cutaneous melanoma patients were included. Geographic trial distribution as well as disease state and type of intervention were analyzed and compared among each group. ClinicalTrials.gov is an invaluable tool to study the research enterprise. Further studies are needed on prevention and early detection of melanoma in the curative setting, a critical role for dermatologists.
Practice Points
- The landscape of melanoma clinical trial research has shifted to follow advances in targeted therapy and immunotherapy.
- With these new treatments there is an increased risk for nonmelanoma skin toxicities requiring increased vigilance and collaboration between dermatologists and oncologists.
- Physicians are encouraged to use ClinicalTrials.gov to find details and contact information for actively recruiting clinical trials and results on completed trials.
Characteristics of these 777 trials were varied. Many interventions were in the early stages of development, with 339 (44%) trials classified as phase 0, phase 1, or phase 1/phase 2; 306 (39%) as phase 2; and 71 (9%) as nonpharmacologic (nonphase) trials. Only 58 trials (8%) were classified as phase 3 or phase 4. The majority of the trials were actively recruiting (225 [29%]), active but not yet recruiting (172 [22%]), or completed (255 [33%]); however, 98 trials (13%) had been suspended, terminated, or withdrawn. Additionally, 22 trials (3%) were not yet recruiting and 5 (<1%) were classified as “other” because they did not have a recruitment status listed.
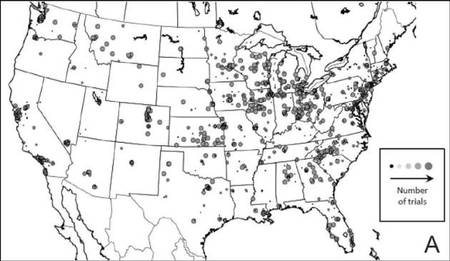
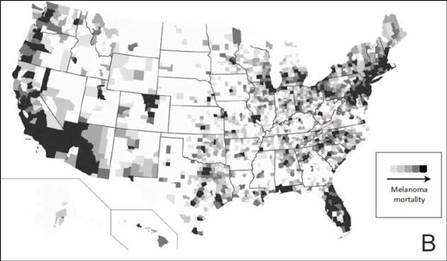
The distribution of actively enrolling clinical trials corresponds to major metropolitan areas within the Northeast, Upper Midwest, and Coastal California (Figure 1A). Figure 1B demonstrates the melanoma-specific mortality across the United States. Areas in the Southwest and Florida shared some of the greatest disease burden.
|
| Figure 1. Geographical representation of US clinical trial enrollment with the number of actively recruiting trials for each unique US zip code presented. The circle size corresponds to the number of trials. The largest circles indicate more than 5 trials within a given zip code (A). County-level melanoma-specific mortality data are presented for 2001 to 2010 (B). Darkest areas represent the highest numbers of melanoma deaths. |
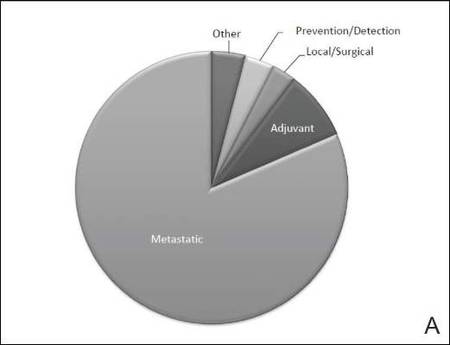
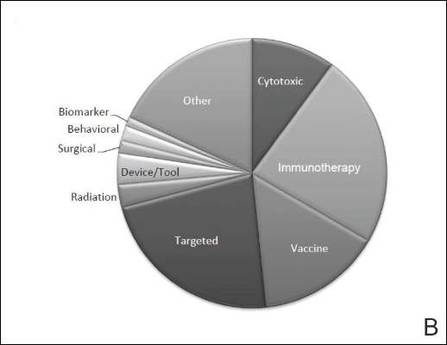
The disease state and type of intervention for all the included trials are summarized in Figure 2. The vast majority of trials (633/777 [82%]) enrolled participants with metastatic melanoma. Unlike many other tumor types, only 64 (8%) trials enrolled patients specifically in the adjuvant setting. Most trials focused on targeted (175 [23%]), immune (180 [23%]), and vaccine (117 [15%]) therapy.
|
| Figure 2. Trial distribution stratified by disease state (A) and type of intervention (B). Trial distribution is shown for 777 interventional clinical trials including melanoma patients. The majority of clinical trials involved patients with metastatic melanoma. The majority of trials investigated targeted therapy, immunotherapy, and vaccine therapy. |
We subsequently analyzed changes in trial characteristics over time. We noted a decrease in the number of trials investigating cytotoxic and vaccine-based therapies, and increasing numbers of trials investigating immunotherapy (P=.041). Between 2005 and 2007, 14% (27/201) of all trials investigated cytotoxic therapies compared to just 7% (20/294) of trials between 2011 and 2013. With the approval of ipilimumab, 29% (85/294) of all clinical trials between 2011 and 2013 investigated immunotherapies, which comprised only 18% (37/201) of clinical trials between 2005 and 2007. The majority of trials continued to enroll patients in the metastatic setting where outcomes remain poor. Importantly, only 6% (49/777) of all clinical trials have focused on prevention, early detection, and local management of melanoma, which has remained constant over time.
Comment
Cutaneous melanoma remains an area of active investigation, interdisciplinary collaboration, and great promise. The ClinicalTrials.gov registry serves not only to increase transparency among interested parties but also as a rich resource to study the clinical research landscape as demonstrated in this study.
Greater understanding of the underlying genetic and immunogenic properties of melanoma tissues has led to the US Food and Drug Administration approval of several novel agents to treat metastatic disease. BRAF inhibitors such as vemurafenib and dabrafenib target more than 50% of all melanoma tumors harboring mutations in the BRAF gene and have shown unparalleled efficacy in clinical trials; however, durability of response and adverse effects still remain a concern.4,9-11 Ipilimumab, a CTLA-4 inhibitor, enhances antitumor immunity and demonstrated improved survival in clinical trials.12,13 Nivolumab, a fully human IgG4 programmed death 1 (PD-1) immune-checkpoint inhibitor antibody, also demonstrated improved overall and progression-free survival.14 Finally, trametinib, a MEK inhibitor, used in combination with BRAF inhibitors has demonstrated improved response over BRAF inhibitors alone.15
Although traditional cytotoxic chemotherapy was one of the few available treatment options before 2011, response was infrequent.16 Our data indicate that the melanoma research landscape has shifted to follow advances in targeted therapy and immunotherapy. We noted a decrease in the study of cytotoxic chemotherapy in metastatic melanoma, with a compensatory increase in immunotherapy trials and a continued commitment to targeted therapy. Further, with the approval of BRAF inhibitors, CTLA-4 inhibitors, and PD-1 inhibitors for metastatic disease, some have pushed to move these agents into the adjuvant setting to prevent micrometastases from evolving into clinically significant disease.17 Early results from EORTC (European Organisation for Research and Treatment of Cancer) 18071 comparing adjuvant ipilimumab to placebo demonstrated a 26.1-month versus 17.1-month improvement in relapse-free survival, respectively.18 However, this finding has important implications for clinical dermatologists. Patients treated with BRAF inhibitors are at increased risk for keratoacanthomas, invasive squamous cell carcinomas, and secondary primary melanomas.19,20 Caring for these patients requires increased vigilance and collaboration between dermatologists and oncologists.






