The role of repeat uterine curettage in postmolar gestational trophoblastic neoplasia
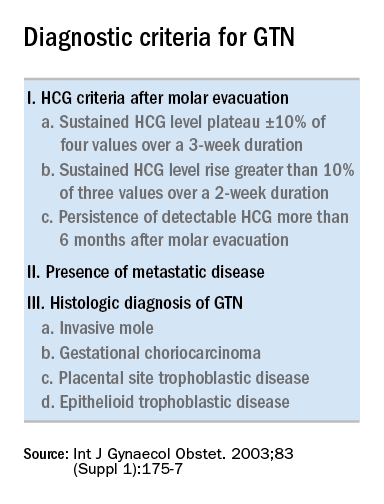
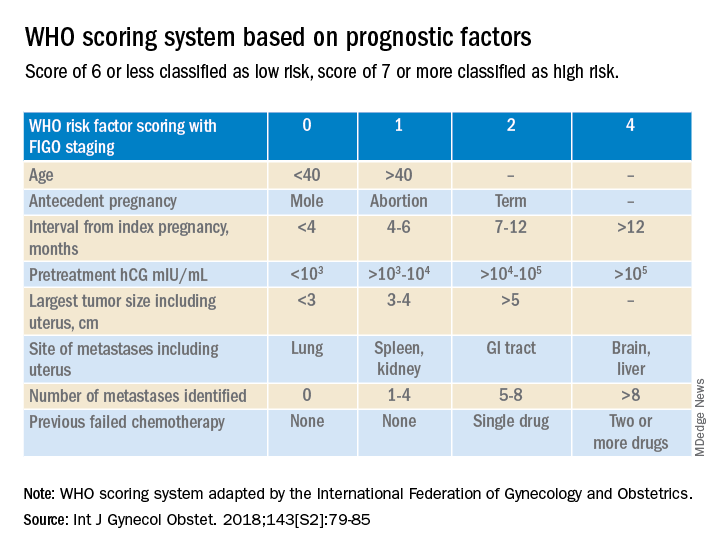
Trophoblastic tissue is responsible for formation of the placenta during pregnancy. Gestational trophoblastic disease (GTD), a group comprising benign (hydatidiform moles) and malignant tumors, occurs when gestational trophoblastic tissue behaves in an abnormal manner. Hydatidiform moles, which are thought to be caused by errors in fertilization, occur in approximately 1 in 1,200 pregnancies in the United States. Gestational trophoblastic neoplasia (GTN) refers to the subgroup of these trophoblastic or placental tumors with malignant behavior and includes postmolar GTN, invasive mole, gestational choriocarcinoma, placental-site trophoblastic tumor (PSTT), and epithelioid trophoblastic tumor. Postmolar GTN arises after evacuation of a molar pregnancy and is most frequently diagnosed by a plateau or increase in human chorionic gonadotropin (hCG).1 The risk of postmolar GTN is much higher after a complete mole (7%-30%) compared with a partial mole (2.5%-7.5%).2 Once postmolar GTN is diagnosed, a World Health Organization score is assigned to determine if patients have low- or high-risk disease.3 The primary treatment for most GTN is chemotherapy. A patient’s WHO score helps determine whether they would benefit from single-agent or multiagent chemotherapy. The standard of care for low-risk disease is single-agent chemotherapy with either methotrexate or actinomycin D.
The role of a second uterine curettage, after the diagnosis of low-risk postmolar GTN, has been controversial because of the limited data and disparate outcomes reported. In older retrospective series, a second curettage affected treatment or produced remission in only 9%-20% of patients and caused uterine perforation or major hemorrhage in 5%-8% of patients.4,5 Given relatively high rates of major complications compared with surgical cure or decreased chemotherapy cycles needed, only a limited number of patients seemed to benefit from a second procedure. On the other hand, an observational study of 544 patients who underwent second uterine evacuation after a presumed diagnosis of persistent GTD found that up to 60% of patients did not require chemotherapy afterward.6 Those with hCG levels greater than 1,500 IU/L or histologic evidence of GTD were less likely to have a surgical cure after second curettage. The indications for uterine evacuations were varied across these studies and make it nearly impossible to compare their results.
More recently, there have been two prospective trials that have tackled the question of the utility of second uterine evacuation in low-risk, nonmetastatic GTN. The Gynecologic Oncology Group performed a single-arm prospective study in the United States that enrolled patients with postmolar GTN to undergo second curettage as initial treatment of their disease.7 Of 60 eligible patients, 40% had a surgical cure (defined as normalization of hCG followed by at least 6 months of subsequent normal hCG values). Overall, 47% of patients were able to avoid chemotherapy. All surgical cures were seen in patients with WHO scores between 0 and 4. Importantly, three women were diagnosed with PSTT, which tends to be resistant to methotrexate and actinomycin D (treatment for nonmetastatic PSTT is definitive surgery with hysterectomy). The study found that hCG was a poor discriminator for achieving surgical cure. While age appeared to have an association with surgical cure (cure less likely for younger and older ages, younger than 19 and older than 40), patient numbers were too small to make a statistical conclusion. There were no uterine perforations and one patient had a grade 3 hemorrhage (requiring transfusion).
In the second prospective trial, performed in Iran, 62 patients were randomized to either second uterine evacuation or standard treatment after diagnosis of postmolar GTN.8 All patients in the surgical arm received a cervical ripening agent prior to their procedure, had their procedure under ultrasound guidance, and received misoprostol afterward to prevent uterine bleeding. Among those undergoing second uterine evacuation, 50% were cured (no need for chemotherapy). Among those needing chemotherapy after surgery, the mean number of cycles of chemotherapy needed (3.07 vs. 6.69) and the time it took to achieve negative hCG (3.23 vs. 9.19 weeks) were significantly less compared with patients who did not undergo surgery. hCG prior to second uterine evacuation could distinguish response to surgery compared with those needing chemotherapy (hCG of 1,983 IU/L or less was the level determined to best predict response). No complications related to surgery were reported.
Given prospective data available, second uterine evacuation for treatment of nonmetastatic, low-risk postmolar GTN is a reasonable treatment option and one that should be considered and discussed with patients given the potential to avoid chemotherapy or decrease the number of cycles needed. It may be prudent to limit the procedure to patients with an hCG less than 1,500-2,000 IU/L and to those between the ages of 20 and 40. While uterine hemorrhage and perforation have been reported in the literature, more recent data suggest low rates of these complications. Unfortunately, given the rarity of the disease and the historically controversial use of second curettage, little is known about the effects on future fertility that this procedure may have, including the development of uterine synechiae.
Dr. Tucker is assistant professor of gynecologic oncology at the University of North Carolina at Chapel Hill.
References
1. Ngan HY et al, FIGO Committee on Gynecologic Oncology. Int J Gynaecol Obstet. 2003 Oct;83 Suppl 1:175-7. Erratum in: Int J Gynaecol Obstet. 2021 Dec;155(3):563.
2. Soper JT. Obstet Gynecol. 2021 Feb.;137(2):355-70.
3. Ngan HY et al. Int J Gynecol Obstet. 2018;143:79-85.
4. Schlaerth JB et al. Am J Obstet Gynecol. 1990 Jun;162(6):1465-70.
5. van Trommel NE et al. Gynecol Oncol. 2005 Oct;99(1):6-13.
6. Pezeshki M et al. Gynecol Oncol. 2004 Dec;95(3):423-9.
7. Osborne RJ et al. Obstet Gynecol. 2016 Sep;128(3):535-42.
8. Ayatollahi H et al. Int J Womens Health. 2017 Sep 21;9:665-71.





