Sepsis Presenting in Hospitals versus Emergency Departments: Demographic, Resuscitation, and Outcome Patterns in a Multicenter Retrospective Cohort
BACKGROUND: Differences between hospital-presenting sepsis (HPS) and emergency department-presenting sepsis (EDPS) are not well described.
OBJECTIVES: We aimed to (1) quantify the prevalence of HPS versus EDPS cases and outcomes; (2) compare HPS versus EDPS characteristics at presentation; (3) compare HPS versus EDPS in process and patient outcomes; and (4) estimate risk differences in patient outcomes attributable to initial resuscitation disparities.
DESIGN: Retrospective consecutive-sample cohort.
SETTING: Nine hospitals from October 1, 2014, to March 31, 2016.
PATIENTS: All hospitalized patients with sepsis or septic shock, as defined by simultaneous (1) infection, (2) ≥2 Systemic Inflammatory Response Syndrome (SIRS) criteria, and (3) ≥1 acute organ dysfunction criterion. EDPS met inclusion criteria while physically in the emergency department (ED). HPS met the criteria after leaving the ED.
MEASUREMENTS: We assessed overall HPS versus EDPS contributions to case prevalence and outcomes, and then compared group differences. Process outcomes included 3-hour bundle compliance and discrete bundle elements (eg, time to antibiotics). The primary patient outcome was hospital mortality.
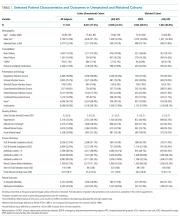
RESULTS: Of 11,182 sepsis hospitalizations, 2,509 (22.4%) were hospital-presenting. HPS contributed 785 (35%) sepsis mortalities. HPS had more frequent heart failure (OR: 1.31, CI: 1.18-1.47), renal failure (OR: 1.62, CI: 1.38-1.91), gastrointestinal source of infection (OR: 1.84, CI: 1.48-2.29), euthermia (OR: 1.45, CI: 1.10-1.92), hypotension (OR: 1.85, CI: 1.65-2.08), or impaired gas exchange (OR: 2.46, CI: 1.43-4.24). HPS were admitted less often from skilled nursing facilities (OR: 0.44, CI: 0.32-0.60), had chronic obstructive pulmonary disease (OR: 0.53, CI: 0.36-0.78), tachypnea (OR: 0.76, CI: 0.58-0.98), or acute kidney injury (OR: 0.82, CI: 0.68-0.97). In a propensity-matched cohort (n = 3,844), HPS patients had less than half the odds of 3-hour bundle compliant care (17.0% vs 30.3%, OR: 0.47, CI: 0.40-0.57) or antibiotics within three hours (66.2% vs 83.8%, OR: 0.38, CI: 0.32-0.44) vs EDPS. HPS was associated with higher mortality (31.2% vs 19.3%, OR: 1.90, CI: 1.64-2.20); 23.3% of this association was attributable to differences in initial resuscitation (resuscitation-adjusted OR: 1.69, CI: 1.43-2.00).
CONCLUSIONS: HPS differed from EDPS by admission source, comorbidities, and clinical presentation. These patients received markedly less timely initial resuscitation; this disparity explained a moderate proportion of mortality differences.
© 2019 Society of Hospital Medicine
Database Design and Structure
The Northwell Sepsis Database has previously been described in detail.11,13,14 Briefly, all patients met clinical sepsis criteria: (1) infection AND (2) ≥2 (SIRS) criteria AND (3) ≥1 acute organ dysfunction criterion. Organ dysfunction criteria were hypotension, acute kidney injury (AKI), coagulopathy, altered gas exchange, elevated bilirubin (≥2.0 mg/dL), or altered mental status (AMS; clarified in Supplemental Table 1). All organ dysfunction was not otherwise explained by patients’ medical histories; eg, patients on warfarin anticoagulation were not documented to have coagulopathy based on international normalized ratio > 1.5. The time of the sepsis episode (and database inclusion) was the time of the first vital sign measurement or laboratory result where a patient simultaneously met all three inclusion criteria: infection, SIRS, and organ dysfunction. The database excludes patients who were <18 years, declined bundle interventions, had advance directives precluding interventions, or were admitted directly to palliative care or hospice. Abstractors assumed comorbidities were absent if not documented within the medical record and that physiologic abnormalities were absent if not measured by the treatment team. There were no missing data for the variables analyzed. We report analysis in adherence with the STROBE statement guidelines for observational research.
Exposure
The primary exposure was whether patients had EDPS versus HPS. We defined EDPS patients as meeting all objective clinical inclusion criteria while physically in the ED. We defined HPS as first meeting sepsis inclusion criteria outside the ED, regardless of the reason for admission, and regardless of whether patients were admitted through the ED or directly to the hospital. All ED patients were admitted to the hospital.
Outcomes
Process outcomes were full 3-hour bundle compliance, time to antibiotic administration, blood cultures before antibiotics, time to fluid initiation, the volume of administered fluid resuscitation, lactate result time, and whether repeat lactate was obtained (Supplemental Table 2). Treatment times were times of administration (rather than order time). The primary patient outcome was hospital mortality. Secondary patient outcomes were mechanical ventilation, ICU admission, ICU days, hospital length of stay (LOS). We discounted HPS patients’ LOS to include only days after meeting the inclusion criteria. Patients were excluded from the analysis of the ICU admission outcome if they were already in the ICU prior to meeting sepsis criteria.
Statistical Analysis
We report continuous variables as means (standard deviation) or medians (interquartile range), and categorical variables as frequencies (proportions), as appropriate. Summative statistics with 95% confidence intervals (CI) describe overall group contributions. We used generalized linear models to determine patient factors associated with EDPS versus HPS, entering random effects for individual study sites to control for intercenter variability.
Next, to generate a propensity-matched cohort, we computed propensity scores adjusted from a priori selected variables: age, sex, tertiary versus community hospital, congestive heart failure (CHF), renal failure, COPD, diabetes, liver failure, immunocompromise, primary source of infection, nosocomial source, temperature, initial lactate, presenting hypotension, altered gas exchange, AMS, AKI, and coagulopathy. We then matched subjects 1:1 without optimization or replacement, imposing a caliper width of 0.01; ie, we required matched pairs to have a <1.0% difference in propensity scores. The macro used to match subjects is publically available.15
We then compared resuscitation and patient outcomes in the matched cohort using generalized linear models, ie, doubly-robust estimation (DRE).16 When assessing patient outcomes corrected for resuscitation, we used mixed DRE/multivariable regression. We did this for two reasons: first, DRE has the advantage of only requiring only one approach (propensity vs covariate adjustments) to be correctly specified.16 Second, computing propensity scores adjusted for resuscitation would be inappropriate given that resuscitation occurs after the exposure allocation (HPS vs EDPS). However, these factors could still impact the outcome and in fact, we hypothesized they were potential mediators of the exposure effect. To interrogate this mediating relationship, we recapitulated the DRE modeling but added covariates for resuscitation factors. Resuscitation-adjusted models controlled for timeliness of antibiotics, fluids, and lactate results; blood cultures before antibiotics; repeat lactate obtained, and fluid volume in the first six hours. Since ICU days and LOS are subject to competing risks bias (LOS could be shorter if patients died earlier), we used proportional hazards models where “the event” was defined as a live discharge to censor for mortality and we report output as inverse hazard ratios. We also tested interaction coefficients for discrete bundle elements and HPS to determine if specific bundle elements were effect modifiers for the association between the presenting location and mortality risk. Finally, we estimated attributable risk differences by comparing adjusted odds ratios of adverse outcome with and without adjustment for resuscitation variables, as described by Sahai et al.17
As sensitivity analyses, we recomputed propensity scores and generated a new matched cohort that excluded HPS patients who met criteria for sepsis while already in the ICU for another reason (ie, excluding ICU-presenting sepsis). We then recapitulated all analyses as above for this cohort. We performed analyses using SAS version 9.4 (SAS Institute, Cary, North Carolina).