Update on Management of Barrett’s Esophagus for Primary Care Providers
From the Gastroenterology and Hepatology Section, Baylor College of Medicine, Houston, TX.
Abstract
- Objective: To provide an update on management of Barrett’s esophagus.
- Methods: Review of the literature.
- Results: Management of Barrett’s esophagus depends on the degree of dysplasia. Surveillance by endoscopy every 3–5 years is recommended in patients with Barrett’s esophagus without dysplasia. Patients with Barrett’s esophagus and low-grade dysplasia should undergo surveillance by endoscopy in 3 months for confirmation of the diagnosis; if the diagnosis is confirmed then surveillance by endoscopy or eradication of Barrett’s epithelium by ablation or endoscopic resection are recommended. There is a sufficient evidence to recommend radiofrequency ablation of high-grade dysplasia within Barrett’s esophagus or to perform endoscopic mucosal resection of nodular Barrett’s esophagus with any degree of dysplasia. Early esophageal cancers that are limited to the mucosa can be treated by endoscopic resection, while cancer invading into the deep submucosa or muscularis propria may need esophagectomy with or without chemoradiation.
- Conclusion: The management of Barrett’s esophagus depends on the degree of dysplasia. Radiofrequency ablation and endoscopic mucosal resection are the most commonly used treatment for Barrett’s esophagus with dysplasia.
Keywords: Barrett’s esophagus; radiofrequency ablation; endoscopic submucosal dissection; endoscopic mucosal resection; early esophageal cancer.
Barrett’s esophagus is a common complication of chronic reflux disease [1]. Metaplastic changes that occur at the distal esophageal epithelium are usually asymptomatic [2,3] and occur as reparative adaptations to the insult of the gastric acid [4]. The management of Barrett’s esophagus after diagnosis is currently debated amongst experts without a clear consensus [5,6]. This review is generally consistent with the 2016 guidelines from the American College of Gastroenterology [1], a 2012 guideline from the American Society of Gastrointestinal Endoscopy [7], a 2011 guideline, and a 2016 expert review from the American Gastroenterological Association [8,9].
D efinition
Barrett’s esophagus is a metaplasia of the stratified squamous epithelium to a specialized columnar intestinal epithelium of mucus cells and goblet cells at the distal esophagus secondary to gastroesophageal reflux disease (GERD) [7,10]. Barrett’s esophagus is unstable tissue which can progress to esophageal adenocarcinoma. When unmanaged, the risk of cancer in dysplastic mucosa is at least thirty-fold greater than that for the general population [11–14], with recent studies suggesting a 0.4–0.7 occurrence rate per year [11,15]. With no dysplasia, the risk is low [16].
Epidemiology
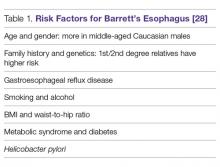
The prevalence of Barrett’s esophagus is ~10% in patients with GERD [11–13,17], an estimate tied to the prevalence of GERD. However, due to the lack of symptoms of Barrett’s esophagus, no solid data supports this assumption [18–20]. A European study estimated the prevalence of Barrett’s esophagus to be 1.6% among the general population [21,22]. Barrett’s esophagus is usually diagnosed during endoscopic examinations of middle-aged and older adults, with the mean age being 55 years of age. It is most commonly found in Caucasian males and associated with the use of smoking tobacco. The male-to-female ratio is approximately 2:1 [1] and it appears to be uncommon in African Americans [23,24]. Abdominal obesity as measured by an increased waist-to-hip ratio is associated with an increased risk of Barrett’s esophagus [25,26]. Germline mutations in the MSR1, ASCC1, and CTHRC1 genes have been associated with the presence of Barrett’s esophagus and esophageal adenocarcinoma [27]. Risk factors are listed in Table 1.
Clinical Symptoms
Columnar metaplasia itself does not cause any symptoms but is merely the adaptation of the cells to the repeated effect of the acid. The main clinical symptoms of the disease would initially be symptoms associated with GERD, such as heartburn, water brash, and dysphagia [1]. Severe presentations of GERD, such as esophageal ulceration, stricture, and hemorrhage, usually occur with long-segment Barrett’s esophagus [29,30]. However, 40% of patients presenting with adenocarcinoma had no history of GERD or symptoms of heartburn [13]. Furthermore, as few as 5% of those presenting with adenocarcinoma were known to have Barrett’s esophagus [31].
D iagnosis
Barrett’s esophagus generally requires an endoscopic examination with biopsy confirmation from the distal esophagus showing specialized intestinal columnar epithelium [32]. The biopsy specimen is acquired from the cellular lining proximal to gastroesophageal junction [5,33]. Barrett’s esophagus is classified into long- and short-segment based on the length of salmon-colored mucosa in the distal esophagus. A distance longer than 3 cm is
The Prague classification was presented by an international research group in 2006 and is regarded as the standard for measuring the length of Barrett’s esophagus. The lower measurement boundary is formed by the proximal cardial notch, and the 2 upper measurement boundaries are marked by the proximal limit of the circumferential Barrett’s segment and the longest tongue of Barrett’s [37].Confirmation of the diagnosis of dysplastic
Once the initial diagnosis of Barrett’s esophagus is made, we recommend referring the patient to a Barrett’s esophagus specialized center in order to offer the patient a second opinion from a team of experts in Barrett’s esophagus. This would avoid possible false-positive results, which can be as high as 40% [1,8]. It would also offer the patient a comprehensive multidisciplinary approach and adequate long-term management. It is further preferred if an advanced intervention is offered to the patient such as endoscopic mucosal resection or endoscopic submucosal dissection. Expert endoscopists
The availability of advanced endoscopic tools improves diagnostic yield. Adopting advanced techniques can reduce errors during biopsy sampling [40–42]. Some of the newer advanced imaging endoscopic tools include chromoendoscopy, optical coherence tomography, confocal microendoscopy, autofluorescence endoscopy, narrow band imaging (NBI), and Fujinon intelligent chromoendoscopy (FICE) [43,44]. In a meta-analysis examining whether advanced techniques improved diagnostic yield, it was found that advanced imaging increased the diagnostic yield by 34%. Advanced technology mentioned here is not mandated in current guidelines [5,45].
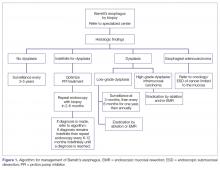
Histopathology categories and TNM staging system for Barrett’s esophagus are shown in Table 2 and Table 3. A management algorithm based on histologic findings is presented in Figure 1.