Treatments for Obstructive Sleep Apnea
Positional Therapy
It has been known for some time that sleeping in a supine position doubles a patient’s AHI compared to sleeping in the lateral position [39]. A more recent analysis showed that 60% of patients were “supine predominant OSA;” these patients had supine AHI that was twice that of non-supine AHI [40]. Moreover, a drug-induced sleep endoscopy study showed that the upper airway collapses at multiple levels sleeping in the supine position as opposed to at a single level sleeping in the lateral position [41]. Another study showed that lateral sleeping position improved passive airway anatomy and decreased collapsibility [42]. Many studies have shown that patients who wear a device that alerts the sleeper that he or she is in a supine position (referred to as positional therapy) significantly decreases AHI, but long-term compliance is still an issue, and new and improved devices are needed [43]. Three new studies bolster the effectiveness of positional therapy [44–46]. In all 3 studies, sleeping in the supine position went down to 0% (no change in sleep efficiency [the ratio of total time spent sleeping to the total time spent in bed]), AHI decreased to less than 6, and sleep quality and daytime sleepiness increased and decreased, respectively [44–46]. Compliance was as low as 76% [44] and as high as 93% [46]. For those who cannot tolerate CPAP, positional therapy could be a substitute for decreasing severity of OSA. However, “phenotyping” OSA patients as “supine predominant OSA” would need to be implemented to guarantee efficacy of positional therapy.
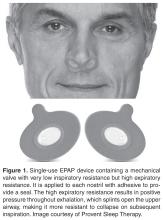
Nasal Expiratory Positive Airway Pressure
Oral Pressure Therapy
Retro-palatal collapse occurs in OSA and can be prevented by applying negative pressure to the upper airway [49]. The oral pressure therapy (OPT) device applies gentle suction anteriorly and superiorly to displace the tongue and soft palate and breathing occurs via nasopharyngeal airway [12]. A recent systematic review [49] of OPT revealed that successful OPT treatment rate was 25% to 37% if using standard and stringent definitions of treatment success. Although OPT decreased AHI, residual AHI still remained high due to positional apneas and collapse of upper airway at other levels besides retro-palatal. The authors of this systematic review recommend more rigorous and controlled studies with defined “treatment success” [49]. The advantage of OPT is that adherence was good; patients used the device on average 6 hours a night. There were no severe or serious adverse events with OPT, however oral tissue discomfort or irritation, dental discomfort, and dry mouth were reported [50].
Oral Appliances
Similar to OPT, oral appliances (OAs) attempt to prevent upper airway collapse. OAs either stabilize the tongue, advance the mandible, or lift the soft palate to increase the volumes of the upper airways to avert OSA [16, 51]. The OAs, like the mandibular advancement device, for example, have the added benefit of being fitted specifically for the OSA patient. The mandible for a patient can be advanced to alleviate obstructive apneas, but can also be pulled back if the OA is too uncomfortable or painful. However, there is still dispute on how exactly to titrate these OAs [52]. A meta-analysis recently published looked at all clinical trials of OAs through September 2015. After meeting strict exclusion/inclusion criteria, 17 studies looking at OAs were included in the meta-analysis. There were robust decreases in AHI and in symptoms of sleepiness in OSA patients that used OAs compared to control groups. However, due to the strict inclusion/exclusion criteria of the meta-analysis, all the studies except one used mandibular advancement appliances; one study used a tongue-retaining appliance. The authors concluded that there is sufficient evidence for OAs to be effective in patients with mild-to-moderate OSA [51]. Since the meta-analysis, 6 new studies have been published about OAs. In 4 of the studies (all using mandibular advancement), OAs significantly decreased AHI by 50% or more in the majority of OSA patients [53–56]. The other 2 studies looked at long-term efficacy and compliance. In both studies, there were drastic decreases in AHI when OAs were applied [57, 58]. In one study, about 40% of OSA patients stopped using the OAs. When the change in AHI was stratified between users and non-users, the users group was significantly higher that the non-user group, suggesting that the non-user group were not compliant due to less of an effect of the OA on AHI [57]. In the second study, OSA patients using OAs for a median of 16.5 years were evaluated for long-term efficacy of the OAs. At the short-term follow-up, AHI decreased by more than 50% with use of an OA. However, at the long-term follow-up, the OA lost any effect on AHI. One reason for this is that the OSA patients’ AHI without the OA at the long-term follow-up nearly doubled compared to AHI without OA at the short-term follow-up. The authors conclude that OSA patients using OAs for the long-term might undergo deteriorations in treatment efficacy of OAs, and that regular follow-up appointments with sleep apnea recordings should be implemented [58].
A similarity in all these studies is that adherence was higher for OAs compared to CPAP [51]. The caveat is that most studies have relied on self-reports for adherence rates [12]. However, there were 3 studies that implemented a sensor that measured adherence and compared those results to self-reported OA adherence. All 3 studies showed a strong relationship between self-reports and sensor adherence; patients were honest about adherence to OAs [59–61]. Studies have also been conducted to predict compliance with OAs: higher therapeutic CPAP pressure, age, OSA severity [62], decreased snoring [63], and lower BMI [64, 65] predicted compliance, while dry mouth [63], oropharyngeal crowding [65], and sleeping in a supine position [66] predicted noncompliance. Though adherence rates are high, OAs do not decrease AHI as much as CPAP [67], and a recent study showed that long-term adherence rates might not be different to CPAP adherence rates [68]. OAs, due to their higher adherence rates, are a potential second choice over CPAP. However, they are less efficacious than CPAP at decreasing AHI. That may not be as important since a recent meta-analysis comparing the effects of CPAP or OAs on blood pressure showed that both treatments significantly decreased blood pressure [69]. More studies need to be conducted over long-term efficacy of OAs compared with CPAP.
Surgeries to Treat OSA
Surgery as a treatment option has been extensively reviewed and meta-analyzed [70–78]. Surgery for the treatment of OSA includes tongue suspension [70,74], maxillomandibular advancement (MMA) [72,73,78], pharyngeal surgeries (eg, uvulopharyngopalatoplasty [UPPP]) [73], laser-assisted uvulopalatoplasty (LAUP) [73], radiofrequency ablation (RFA) [73], tracheostomy [71], nasal surgery [75], and glossectomy [77], as well as multi-level and multi-phased procedures [70,74,76,77]. Most studies done on surgeries were case studies, with a minority of investigations that were randomized and controlled. Glossectomy, as part of a multi-level surgical approach, decreased AHI and symptoms of sleepiness, but glossectomy as a stand-alone surgical procedure did not improve AHI [77]. Significant improvements in AHI and sleepiness symptoms were seen in a majority of OSA patients who underwent MMA [72,73,78] and tracheostomy, although tracheostomy was performed for the morbidly obese or those who have failed other traditional surgical treatments [71]. Stand-alone tongue suspension and nasal surgery did not decrease AHI in the majority of patients, though nasal surgery did decrease subjective sleepiness [70,72,74,75]. However, tongue suspension combined with UPPP had better outcomes [70]. LAUP showed inconsistent results with the majority of studies showing no change in AHI, while UPPP and RFA seemed to improved AHI, although some studies showed no change [73]. Multi-level or multi-phase surgeries also showed improvements on OSA severity, but whether these surgeries are better than stand-alone remains to be investigated [73,76]. Morbidity and adverse events, like infection or pain, are common in all of these surgical events [70–78], but there are significant differences between the procedures. For example, MMA had fewer adverse events reported compared to UPPP [73]. More recently, glossectomy via transoral robotic surgery with UPPP [79] or epiglottoplasty [80] has been investigated; there were decreases in AHI, but response rates were between 64% to 73%. Although it seems surgical procedures to treat OSA are plausible, most studies were not rigorous enough to say this with any certainty.
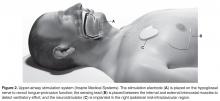
Hypoglossal Nerve Stimulation
OSA subjects experience upper airway obstruction due to loss of genioglossus muscle activity during sleep. Without tongue activation, the negative pressure of breathing causes the upper airways to collapse [81]. Transcutaneous, intraoral, and intramuscular devices used to electrically activate the tongue have been developed and tested; however, although these devices decreased AHI they also induced arousals and sleep fragmentation caused by the electrical stimulus [82–86]. A new method had to be developed that would not be felt by the OSA patient.
In all trials to date, there were significant decreases in AHI as long as 3 years post implantation [87–93]. There were significant improvements in symptoms of sleepiness, mood, quality of life, and sleep quality [87,88,90–94]. When OSA patients had their neurostimulators turned off for 5 days to a week, AHI returned back to baseline levels [89,92]. However, all the trials excluded morbidly obese individuals [87–93] because investigations showed that HNS had no therapeutic effect with elevated BMI [88,90]. The drawbacks of HNS are that it is surgically invasive and minor adverse events have been reported: procedural-related events (eg, numbness/pain/swelling/infection at incision site, temporary tongue weakness) that resolved with time, pain medication, and/or antibiotic treatment, or therapy-related events (eg, tongue abrasions cause by tongue movement over teeth, discomfort associated with stimulation) that resolved after acclimation. Serious adverse events occurred infrequently, such as infection at incision site requiring device removal or subsequent surgery to reposition or replace electrode cuff or malfunctioning neurostimulator [87,88,90]. HNS durability at 18 and 36 months was still very effective, with decreased AHI and increase quality of life and sleep being sustained; adverse events were uncommon this long after implantation [91,93]. Although surgery is required and adverse events are reported, the long-term significant improvement of OSA makes this a very viable treatment option over CPAP. However, increasing prevalence rates of OSA are correlated to increasing obesity rates [2], which may limit the usefulness of HNS since a BMI of more than 40 might preclude individuals to this treatment.
Pharmacologic Treatment
There are no approved pharmacologic treatments for OSA. A recent Cochrane review and meta-analysis assessed clinical trials of various drugs treating OSA. These drugs targeted 5 strategies at alleviating OSA: increasing ventilatory drive (progestogens, theophylline, and acetazolamide), increasing upper airway tone (serotonergics and cholinergics), decreasing rapid eye movement sleep (antidepressants and clonidine), increasing arousal threshold (eszopiclone), and/or increasing the cross-sectional area or reducing the surface tension of the upper airway through topical therapy (fluticasone and lubricant). The review concluded that “some of the drugs may be helpful; however, their tolerability needs to be considered in long-term trials.” Some of these drugs had little or no effect on AHI, and if they did have an effect on AHI, side effects outweighed the benefit [95]. Since then, more investigations of other drugs targeted at the previously aforementioned strategies or via new strategies have been published.
Dronabinol (synthetic Δ9-THC), a nonselective cannabinoid type 1 and type 2 receptor agonist, significantly reduced AHI and improved subjective sleepiness and alertness in a single-blind dose-escalation (2.5, 5, or 10 mg) proof-of-concept study [96,97]. Dronabinol most likely increases upper airway tone though inhibition of vagal afferents [98,99]. There were no serious adverse events associated with dronabinol. Minor adverse events included somnolence and increased appetite. Increased appetite might lead to increased weight and contradict any beneficial effects of dronabinol; however, in the 3-week treatment period no weight gain was observed [97]. This might have been due to drug administration occurring before going to sleep with no opportunity to eat. A larger randomized controlled study will be needed to establish the safety and efficacy of dronabinol.
The sedative zopiclone was used to increase arousal threshold without effecting genioglossus activity [100]. Eszopiclone, a drug in the same class, has been used in the past with unfavorable results [95]. Zopiclone was used in a small double-blind randomized controlled cross-over study. Zopiclone significantly increased respiratory arousal threshold without effecting genioglossus activity or the upper airway’s response to negative pressure. Thus, there was a trend in the zopiclone treatment to increase sleep efficiency. However, zopiclone had no effect on AHI, and increased oxygen desaturation [100]. Similar to eszopiclone, the results for zopiclone are not promising.
A new strategy to treat OSA is to modify pharmacologically “loop gain,” a dimensionless value quantifying the stability of the ventilatory control system. A high loop gain signifies instability in the ventilatory control system and predisposes an OSA person to recurrent apneas [101–103]. Three studies used drugs that inhibit carbonic anhydrase to stabilize the ventilatory control system [104–106]. Two studies used acetazolamide, which decreased loop gain in OSA patients [104,105]. Acetazolamide only decreased AHI in non–rapid eye movement (NREM) sleep, and there was a slight correlation between decrease in loop gain and total AHI [105]. Acetazolamide also decreased ventilatory response to spontaneous arousal, thus promoting ventilatory stability [104]. In the last study, zonisamide, a carbonic anhydrase inhibitor that also causes weight loss, was investigated in OSA patients. Sleep apnea alleviation, measured in terms of absolute elimination of sleep apnea by mechanical or pharmacologic treatment, was 61% and 13% for CPAP and zonisamide, respectively, compared with placebo. In other words, zonisamide decreased AHI but not to the extent of CPAP [106]. Zonisamide also decreased arousals and marginally, but significantly, decreased weight compared to the CPAP group. Although carbonic anhydrase inhibitors have promise as an alternative treatment, long-term use is poorly tolerated [101] and further studies need to be completed.
OSA has been linked with gastroesophageal reflux disease (GERD), with studies suggesting OSA precipitates GERD [107] or GERD precipitates OSA [108]. A meta-analysis was recently published looking at studies that used proton pump inhibitors (PPI) to treat GERD and the effects it would have on OSA [109]. The meta-analysis only included 2 randomized trials and 4 prospective cohort studies. Two of the cohort studies showed a significant decrease, and one cohort showed no difference in apnea indices; and all 4 of the cohort studies showed no difference in AHI. In one trial, the frequency of apnea attacks as recorded by diaries significantly decreased. In 3 cohort studies and one trial, symptoms of sleepiness significantly decreased [109]. A study that was not included in the meta-analysis showed that 3 months of PPI treatment decreased AHI but did not alter sleep efficiency [110]. Larger randomized controlled studies need to be conducted on the effects of PPIs on OSA, especially since PPIs are well tolerated with only weak observational associations between PPI therapy and fractures, pneumonia, mortality, and nutritional deficiencies [111].
The drugs mentioned above have potential for treating OSA in patients intolerant to CPAP. The efficacy and side effects of the drugs will need to be studied for long-term use. However, development of pharmacologic treatments has been hampered by incomplete knowledge of the relevant sleep-dependent peripheral and central neural mechanisms controlling ventilatory drive and upper airway muscles. More importantly, additional basic science research needs to focus on the neurobiological and neurophysiological mechanisms underlying OSA to develop new pharmacotherapies or treatment strategies, or to modify previous treatment strategies.
Treatment Combinations and Phenotyping
It has been recently suggested that combining 2 or more of the above treatments might lead to greater decreases in AHI and greater improvements in subjective sleepiness [112,113]. In fact, one such treatment combination has occurred [114]. Both OA or positional therapy decrease AHI. However, the combination of an OA and positional therapy led to further significant decreases in AHI compared to when those treatments were used alone [114]. To correctly combine treatments, the patient will have to be “phenotyped” via polysomnography to discern the specific pathophysiology of the patient’s OSA. There are published reports of methods to phenotype patients according to their sleep positon, ventilation parameters, loop gain, arousal threshold, and upper airway gain, and if apneic events occur in REM or NREM sleep [40,115]. Defining these traits for individual OSA patients can lead to better efficacy and compliance of combination treatments for OSA. Combination treatment coupled with phenotyping are needed to try to reduce AHI to levels achieved with CPAP.
Conclusion
CPAP is the gold standard treatment because it substantially decreases the severity of OSA just by placing a mask over one’s face before going to sleep. However, it is not tolerable to continually have air forced into your upper airways, and new CPAP devices that heat and humidify the air, and auto titrate the pressure, have been developed to increase adherence rates, but with limited success. For all the treatments listed, a majority do not decrease the severity of OSA to levels achieved with CPAP. However, adherence rates are higher and therefore might, in the long-term, be a better option than CPAP. Some treatments involve invasive surgery to open or stabilize the upper airways, or to implant a stimulator, some treatments involve oral drugs with side effects, and some treatments involve placing appliances on your nose or in your mouth. And some treatments can be combined and individually tailored to the OSA patient via “phenotyping.” For all treatments, the benefits and risks need to be weighed by each patient. More importantly, more large randomized controlled studies on treatments or combination of treatments for OSA are needed using parameters such as treatment adherence, AHI, oxygen desaturation, subjective sleepiness, quality of life, and adverse events (both minor and major) to gauge treatment success in the short-term and long-term. Only then can OSA patients in partnership with their health care provider choose the best treatment option.
Corresponding author: Michael W. Calik, PhD, 845 S. Damen Ave (M/C 802), College of Nursing, Room 740, Chicago, IL 60612, mcalik@uic.edu.
Financial disclosures: None.