Global Quest to Cut CAR T Costs
Treatment: Individual, Complex, and Costly
Why is CAR T-cell therapy so expensive? While only a single treatment is needed, the T cells have to go through an “individualized, bespoke manufacturing” process that’s “highly technical,” pediatric oncologist Stephan A. Grupp, MD, PhD, section chief of the Cellular Therapy and Transplant Section at Children’s Hospital of Philadelphia, said in an interview. As he explained, the cells for a single patient have to go through the same testing as with a drug that might be given to 1,000 people.
“The first thing we need to do is collect the cells from a patient,” said Dr. Williams. “For adults, that process involves putting in two big IVs — one in each arm — and then pulling the blood through a machine. This typically involves an 8-hour collection in the hospital and very highly specialized people to oversee the collection process.”
Secondly, at some institutions, “the cells get sent to a company where they undergo the process where the gene is inserted,” she said. “This process needs to be done in a very sterile environment so there’s no infections, and it needs to have a lot of oversight.”
Finally, “after the cells are generated, they are typically frozen and shipped back to the site where the patient is at the hospital,” she said. “Then we give chemotherapy to the patient, which prepares the patient’s blood system. It removes some of the T-cells that are there, allowing for the T cells that we’re about to infuse to quickly be activated, find the cancer, and kill it.”
Side effects can boost costs even more. “Unfortunately, some significant toxicities can occur after we infuse these cells,” Dr. Williams noted. “Patients can have trouble breathing and sometimes need ventilatory support. They can have trouble maintaining their blood pressure and become swollen as fluid seeps into tissues. Or they can have high fevers and organ dysfunction. Many of those patients go to the intensive care unit, which is obviously expensive as well.”
Taking Gene Therapy In-House
As Dr. Williams explained, one way to reduce costs is to “perform the genetic manipulation and expansion of the cells outside of a company.” Several academic institutions in the United States are embracing this approach, including Children’s Hospital of Philadelphia, which is experimenting with an automated device developed by the German company Miltenyi Biotec and known as the CliniMACS Prodigy machine.
“The current manufacturing process is very manual and requires a lot of interaction with the product and highly trained personnel,” Dr. Grupp said. “If you have an automated device, you have those cells in the device over the 7 to 12 days that you actually need to grow the cells. There’s much less interaction, so you need fewer trained personnel.”
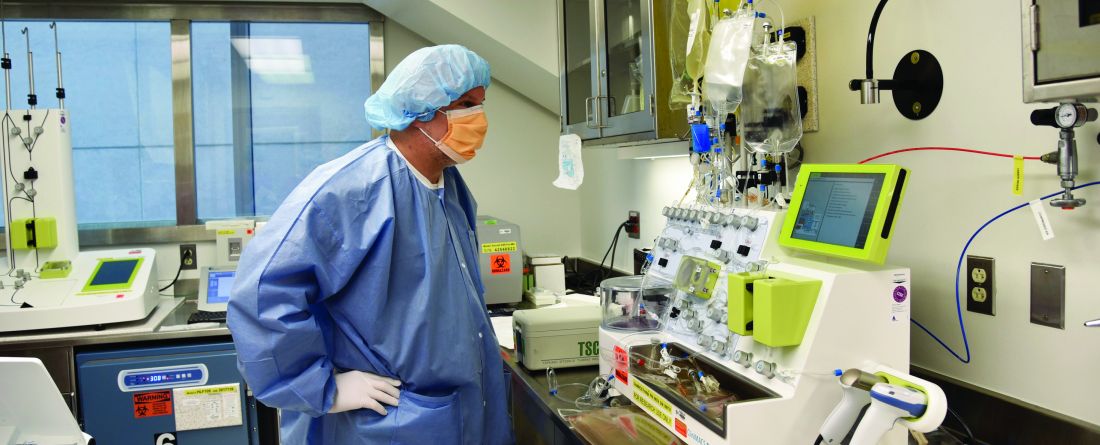
The device is experimental and not yet FDA approved, Dr. Grupp noted, so that patients are all in clinical trials. Children’s Hospital of Philadelphia has treated more than a dozen patients with the device, he said.
Another member of Children’s Hospital of Philadelphia’s CAR T-cell team told WHYY-FM that a single patient’s treatment would run about $30,000 for labor and testing, but not other expenses such as facility costs.
Dr. Grupp estimated that about half a dozen of these devices are in use in the United States, and many more worldwide. “They’re all just like we are — at the absolute beginning. We’ve only been doing this for about a year.”
In the big picture, Dr. Grupp said, “this is where cell therapy is going. Whether it’s point of care or not, automated cell manufacturing is the obvious next step.”






