Evaluation of the Appropriateness of Aspirin Therapy in a Veteran Population
Background: Aspirin is commonly used for primary and secondary prevention of atherosclerotic cardiovascular disease (ASCVD) but may cause more harm than benefit. This study aimed to assess the percentage of patients who were inappropriately prescribed aspirin in the veteran patient population and to assess safety outcomes associated with inappropriate aspirin use.
Methods: Retrospective chart reviews were conducted on up to 200 patients with active prescriptions for 81-mg aspirin tablets filled between October 1, 2019, and September 30, 2021, at the Captain James A. Lovell Federal Health Care Center in Illinois. The primary endpoint was the percentage of patients inappropriately on aspirin therapy and whether these patients were being followed by a clinical pharmacy practitioner. Each patient record was reviewed to determine the appropriateness of aspirin therapy by assessing the indication for use. Safety data were collected for patients who were deemed to be using aspirin inappropriately, including documentation of any major or minor bleeding events.
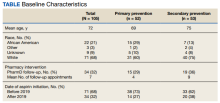
Results: A total of 105 patients were included in this study. For the primary endpoint, 31 patients (30%) had a possible ASCVD risk and were taking aspirin for primary prevention, while 21 patients (20%) had no ASCVD and were taking aspirin for primary prevention. For the secondary endpoint, 25 patients were aged > 70 years, 15 patients were concurrently taking medications that might increase bleeding risk, and 11 patients had chronic kidney disease. Looking at the entire study patient population, for the safety endpoint, 6 patients (6%) experienced a major bleeding event while on aspirin, and 46 (44%) experienced a minor bleeding event while on aspirin.
Conclusions: Common factors seen in this study to warrant deprescribing aspirin for primary prevention included individuals aged > 70 years, concurrent use of medications that increase bleeding risk, and patients with chronic kidney disease. By assessing ASCVD and bleeding risks and having a risk/benefit discussion with patients and prescribers, aspirin used for primary prevention can be appropriately deprescribed when the risks of bleeding outweigh the benefits.
Methods
The objective of this study was to evaluate the appropriateness of aspirin therapy in patient aligned care team (PACT) clinics at the Captain James A. Lovell Federal Health Care Center (FHCC) in North Chicago, Illinois. The PACT clinics are a category of clinics that include all the primary care clinics at FHCC.
The primary outcome of this study was to determine the percentage of patients inappropriately on aspirin therapy. To assess the inappropriate use of aspirin, relevant history of ASCVD was collected. Patients were divided into 3 groups: those with a history of ASCVD, those with no risk factors or history of ASCVD, and those with risk factors and no history of ASCVD. Patients were then categorized for their indication for aspirin use, which included either primary or secondary prevention of ASCVD. Patients were categorized into the primary prevention group if they had no history of ASCVD, whereas patients with a history of ASCVD were placed into the secondary prevention group.
ASCVD was defined as patients with acute coronary syndrome (ACS), history of MI, stable or unstable angina, coronary or other arterial revascularization, stroke, TIA, or peripheral artery disease (PAD), including aortic aneurysm (all with an atherosclerotic origin). Possible ASCVD risk was defined as patients with DM with a major risk factor (family history of premature ASCVD, hypertension, dyslipidemia, smoking, chronic kidney disease [CKD]/albuminuria) or patients diagnosed with coronary artery disease without an event. The percentage of patients followed by a PACT pharmacist, the number of pharmacist follow-up visits during the study period, and the date of the first 81-mg aspirin pharmacy order that was filled at FHCC were also collected.
The secondary outcome of this study focused on patients who were using aspirin for primary prevention and assessed potential reasons that may warrant deprescribing aspirin therapy. One reason for deprescribing is that aspirin may not be indicated for some patients, including those with DM without cardiovascular complications, patients aged > 70 years, and/or patients with CKD (defined as estimated glomerular filtration rate < 60 mL/min). Another reason for deprescribing is contraindication, which included patients with coagulopathy, thrombocytopenia (defined as platelet count < 150,000 mL), a history of gastrointestinal bleeding, peptic ulcer disease or other major bleeds, and/or consistent use of medications that increase bleeding risk (such as nonsteroidal anti-inflammatory agents, steroids, or anticoagulants) for > 14 days.
The safety outcome of this study assessed bleeding events while on aspirin therapy. All patients were categorized depending on if they had a major, minor, or no bleeding event while on aspirin therapy. Hemorrhagic stroke, symptomatic intracranial bleeding, bleeds located in other critical sites or organs (intracranial, intraspinal, intraocular, retroperitoneal, intra‐articular or pericardial), bleeds causing hemodynamic instability requiring vasopressors, bleeds causing a > 2 g/dL hemoglobin drop since initiation of aspirin therapy, severe extracranial bleeding requiring transfusion or hospitalization, fatal bleeding, or bleeds requiring > 2 units of red blood cell transfusion were considered major bleeding events. Minor bleeding events were any events that did not meet the criteria for major bleeding, including bruising, bleeding gums, epistaxis, hemorrhoidal bleeds, and bleeding that did not require intervention or treatment.7
Patients were included if they were aged > 18 years, had an active prescription for 81-mg aspirin tablet on September 30, 2021, and were seen in FHCC PACT clinics or at affiliated community-based outpatient centers. Other doses of aspirin were excluded as the 81-mg dose is the standard dose for primary prevention of ASCVD in the United States. US Department of Defense patients, home-based primary care patients, and community living center patients were excluded in this study. Patients with an aspirin prescription from a non–US Department of Veterans Affairs (VA) facility and patients on aspirin for reasons other than cardiovascular protection (such as pain, fever, etc) also were excluded from this study.
Data were collected from the FHCC electronic health record. A list was generated to include all active prescriptions for aspirin filled at FHCC as of September 30, 2021. Data were reviewed before this date to capture primary and secondary outcomes. No information was gathered from the chart after that date. This project was approved by the Edward Hines, Jr. VA Hospital Institutional Review Board. The primary and secondary outcomes were reported using descriptive statistics.
Results
This study reviewed 140 patient records and 105 patients met inclusion criteria.