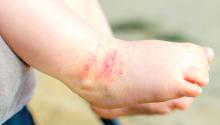
No benefits from bath emollients for childhood eczema
FROM THE BMJ
In a trial of 482 children with atopic dermatitis who were randomized to either use of prescribed emollient bath additives regularly for 12 months or no additives, in addition to usual care, there was no significant difference between the two groups in the patient-oriented eczema measure (POEM) – a score of symptoms in the previous week – for the first 16 weeks of treatment, reported Miriam Santer, MD, of the University of Southampton, England, and her coauthors. The mean POEM score was 7.5 in the bath additives group and 8.4 in the no bath additives group.
There also was no significant effect of emollient bath additives on the secondary outcomes, which included POEM over 52 weeks, dermatitis family impact, generic quality of life, number of eczema exacerbations, or topical corticosteroid or topical calcineurin inhibitor use.
The authors noted that there was the suggestion of benefit in children who bathed five or more times a week, and in children aged under 5 years, but in both cases, the numbers were small.
“Although there is evidence for the need for leave-on emollients and widespread clinical consensus around soap substitutes, there is less agreement about the potential additional benefits of bath additives and a dearth of evidence on their effectiveness,” wrote Dr. Santer and her associates. “Bath additives are, however, widely prescribed at a cost of more than 23 million pounds ($33 million US; 26 million euros) annually to the National Health Service in England.”
The bath emollients used in the study included Aveeno bath oil, and Oilatum bath additive and Balneum bath oil – products available largely in the United Kingdom – along with others. Adherence was reasonable, with 93% of participants in the emollient group using bath additives more than half or all the time, and 92% of those in the no bath additives group using them less than half the time or never.