Psoriasis and Obesity: A Clinical Review of the Bidirectional Link and Management Implications
Psoriasis and obesity are increasingly recognized as comorbid conditions linked by chronic systemic inflammation. This clinical review examines the bidirectional relationship between these 2 diseases, highlighting the shared pathophysiologic pathways involving tumor necrosis factor (TNF) α, interleukin (IL) 6, IL-17, and IL-23. Obesity acts as an independent risk factor for the onset and exacerbation of psoriasis, while psoriatic inflammation promotes metabolic dysregulation. Clinical evidence demonstrates that obesity is associated with greater psoriasis severity, lower quality of life, and a diminished response to systemic therapies, particularly fixed-dose biologics. We discuss the critical role of the dermatologist in early comorbidity screening for metabolic syndrome, liver disease, and psoriatic arthritis. Furthermore, this review evaluates management strategies, emphasizing that weight loss of 5% to 10% can improve cutaneous outcomes and restore therapeutic efficacy. Emerging metabolic therapies, including glucagonlike peptide-1 receptor agonists, represent a promising intersection between dermatologic and metabolic care, with potential benefits for systemic inflammation and psoriasis severity. An integrated treatment approach addressing both inflammatory skin disease and metabolic health is essential for optimal patient care.
Practice Points
- Obesity is an independent risk factor for psoriasis onset and severity. Both conditions share overlapping inflammatory pathways that create a self-perpetuating cycle of metabolic and cutaneous dysfunction.
- Dermatologists play a key role in early detection of comorbidities, and patients with psoriasis and obesity should undergo regular screening for metabolic syndrome, liver disease, and psoriatic arthritis.
- Weight loss is a critical therapeutic intervention that may improve Psoriasis Area and Severity Index scores and restore therapeutic responsiveness.
Psoriasis, a chronic immune-mediated skin disease, is increasingly recognized for its systemic inflammation and associated cardiometabolic risk. The global rise of obesity has revealed a critical link between these 2 conditions. Current evidence confirms that obesity is an independent risk factor that can trigger psoriasis onset, drive more severe disease, and substantially impair treatment efficacy.1,2 This review synthesizes the latest evidence on the shared pathophysiology, clinical consequences, and integrated management strategies for patients with both psoriasis and obesity.
Pathophysiologic Links Between Psoriasis and Obesity
Emerging evidence highlights a bidirectional relationship between psoriasis and obesity rooted in overlapping inflammatory pathways. Both conditions are characterized by chronic inflammation mediated by cytokines that sustain systemic immune activation and metabolic dysfunction. This interplay creates a reciprocal process in which psoriatic inflammation promotes metabolic disturbances while obesity amplifies systemic inflammation and disease severity.3
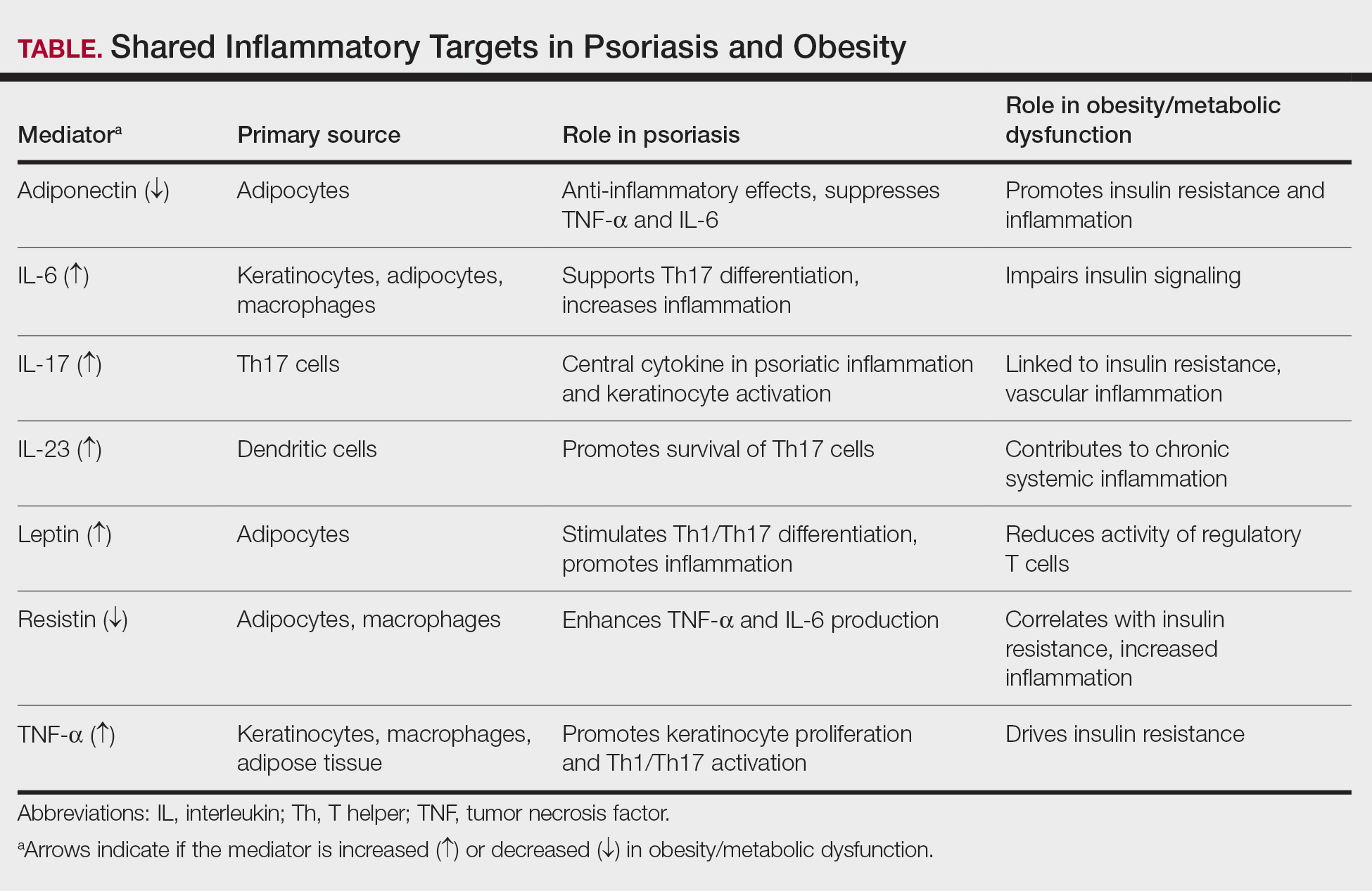
Psoriasis may contribute to obesity through cytokine-driven metabolic alterations in insulin signaling and adipocyte function. The psoriatic immune response is dominated by T helper (Th) 1, Th17, and Th22 cell activation, leading to elevated levels of interferon-γ, tumor necrosis factor (TNF) α, interleukin (IL) 6, IL-17, and IL-22 from keratinocytes.4 These cytokines contribute not only to cutaneous inflammation but also to insulin resistance and adipocyte dysfunction.5 Tumor necrosis factor α and IL-6 interfere with insulin signaling via activation of stress kinases (eg, IκB kinase and c-Jun N-terminal kinase), implicating these molecules in insulin resistance and weight gain.6 Moreover, IL-17, a central cytokine in psoriasis, has been implicated in vascular inflammation, insulin resistance, and type 2 diabetes, suggesting a mechanistic link between psoriatic inflammation and metabolic disease.5 Additionally, chronic systemic inflammation in psoriasis suppresses adiponectin, a protective adipokine that enhances insulin sensitivity and exerts anti-inflammatory effects by inhibiting TNF-α and IL-6 production while promoting IL-10 synthesis. Reduced adiponectin levels have consistently been observed in patients with psoriasis and concomitant obesity or metabolic syndrome.3 The resultant imbalance between proinflammatory and anti-inflammatory mediators creates a metabolic environment conducive to obesity.3
Conversely, obesity itself may intensify both the incidence and severity of psoriasis through shared inflammatory pathways. Leptin, whose expression rises proportionally with adipocyte mass, acts as a proinflammatory mediator linking obesity to psoriasis exacerbation. By promoting Th1 and Th17 cell differentiation and suppressing regulatory T-cell activity, leptin increases IL-17A, IL-6, and TNF-α production.7 These cytokines stimulate keratinocyte proliferation and perpetuate cutaneous inflammation, thereby intensifying disease activity. Similarly, resistin, another adipokine that is elevated in obesity, stimulates monocytes and macrophages to secrete TNF-α and IL-6, creating a proinflammatory state in the body that drives the relationship between excessive fat storage (adiposity) and the development and severity of psoriasis.8 In contrast, reduced adiponectin levels in obesity remove a key anti-inflammatory regulator that normally inhibits TNF-α and IL-6 synthesis and promotes IL-10 production. This deficiency provokes unrestrained cytokine activation within both adipose and cutaneous tissue, exacerbating psoriatic immune dysregulation.9
Free fatty acids derived from abundant adipocytes in obesity further activate nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB)–signaling and induce oxidative stress, augmenting systemic inflammation. Adipose tissue macrophages additionally secrete IL-1Β, IL-6, and TNF-α, which promote keratinocyte proliferation and amplify the psoriatic inflammatory cascade. Importantly, anti–TNF-α therapy has been shown to improve metabolic parameters, reducing low-density lipoprotein and total cholesterol levels and enhancing insulin sensitivity, highlighting the reciprocal benefits of dampening the inflammatory signaling from TNF-α.5
Together, these findings highlight that psoriasis and obesity are interconnected inflammatory disorders driven by overlapping cytokine networks, most notably TNF-α, IL-6, IL-17, and IL-23, and by imbalances in adipokines such as leptin, resistin, and adiponectin (Table).
Clinical Implications of Obesity-Related Comorbidities
Higher body mass index (BMI) has been associated with an increased and earlier incidence of psoriasis. When treated as continuous variables, both Psoriasis Area and Severity Index (PASI) and Dermatology Life Quality Index scores were positively correlated with increases in BMI.10,11 This close association suggests BMI may function as a practical indicator of disease severity and prognosis.7,8,11
Psoriatic arthritis (PsA) is a major comorbidity of psoriasis that impacts disease severity and quality of life. Obesity is associated with an increased risk for PsA after accounting for traditional risk factors. Psoriatic arthritis can cause chronic mobility issues and complicate a patient’s ability to stay active. Early rheumatologist involvement may be warranted to prevent PsA development in patients with obesity and psoriasis.11,12
Patients with psoriasis have an increased baseline risk for metabolic syndrome, including obesity, type 2 diabetes, hypertension, and dyslipidemia11,13,14; therefore, the presence of obesity warrants regular metabolic evaluation. Unmanaged metabolic syndrome contributes to the increased prevalence of myocardial infarction, stroke, and heart failure seen with psoriasis.13,14 Obesity also may cause obstructive sleep apnea, which can exacerbate hypertension and heart disease due to chronic hypoxia.11 The interplay of these metabolic factors puts patients with psoriasis and obesity at heightened cardiovascular risk.13,14
Both psoriasis and obesity present psychosocial challenges such as elevated rates of depression, anxiety, and body-image concerns, all of which become more pronounced when these conditions coexist. These psychological stressors may hinder a patient’s motivation for lifestyle changes or treatment adherence.13
Dermatology visits serve as an important opportunity to monitor obesity-related comorbidities.13,15 Dermatologists are uniquely positioned to initiate metabolic screening while collaborating with primary care physicians for ongoing cardiometabolic management. Metabolic and cardiovascular baselines should be measured when care is established and should be followed longitudinally—these include BMI, waist circumference measurements, blood pressure, lipid panels, fasting glucose or A1C levels, and liver enzymes.10,14,15 Regular screening for depression, suicidality, and disordered eating also is encouraged.13 Clinicians should follow established guidelines to identify and manage metabolic, cardiovascular, and psychological comorbidities.13
Impact of Obesity on Psoriasis Treatment
Obesity is a critical factor in clinical decision-making, as it consistently is associated with diminished response to numerous systemic psoriasis therapies. This reduced efficacy has been observed with conventional oral agents such as methotrexate and cyclosporine and is particularly well documented in the context of biologic therapies.15,16 Several meta-analyses and large real-world studies have shown that higher BMI is associated with a suboptimal treatment response, with patients in higher BMI categories achieving lower rates of PASI 75 and PASI 90 than their nonobese counterparts receiving the same regimen.15,17
This efficacy gap often is rooted in pharmacokinetic challenges. For many biologics administered via subcutaneous injection, increased BMI can lead to altered drug distribution, such as sequestration in adipose tissue. Altered distribution combined with potentially increased drug clearance can result in lower overall serum drug concentrations and subsequent underdosing for a patient’s inflammatory burden.15 This reality highlights the important distinction between fixed and weight-based dosing strategies. Therapies dosed by weight, such as infliximab, have demonstrated more consistent efficacy in populations with obesity, as the dose is escalated to match patient size.18 Despite the weight-dependent effect, recent real-world studies have suggested that fixed dosing of some IL-17A inhibitors (eg, ixekizumab) remains highly effective across BMI categories, while others (eg, secukinumab) show diminished efficacy in obese patients.16,19 Furthermore, some real-world studies have reported an inverse relationship between elevated BMI and efficacy of IL-23 inhibitors, particularly guselkumab, while other studies reported no association.20,21 These mixed observations support a nuanced interpretation of BMI’s role in treatment modification; consideration should be given to the specific medication and the dosing strategy over biologic class alone.
Whether obesity independently drives psoriasis severity or mainly diminishes treatment efficacy through pharmacokinetic effects remains debated. Observational studies show a dose-dependent relationship between BMI and disease severity even in untreated patients, supporting a proinflammatory role for obesity.10 Concurrently, higher BMI may predict lower responses to some fixed-dose biologics, likely due to altered distribution and sequestration in adipose tissue.15,20 The precise contributions are challenging to delineate; both likely converge to heighten disease severity and reduce treatment response.
Beyond efficacy, obesity compounds the safety considerations of systemic treatments. Psoriasis and obesity are both strong independent risk factors for metabolic dysfunction–associated steatotic liver disease. This prevalent comorbidity creates a substantial clinical dilemma, as a first-line, cost-effective agent such as methotrexate carries a known risk for hepatotoxicity, which is amplified in patients with pre-existing liver steatosis.13 Consequently, the presence of obesity and metabolic dysfunction–associated steatotic liver disease often limits the use of methotrexate, forcing a change in therapy. Thorough baseline comorbidity screening in all patients with psoriasis and obesity is necessary to select a therapy that balances efficacy with safety.13,14
Management Strategies
Weight management plays a pivotal role in improving psoriasis outcomes. Even modest weight loss of 5% to 10% has been shown in randomized and observational studies to substantially reduce disease severity, reflected by lower PASI and Dermatology Life Quality Index scores and enhance treatment responsiveness.22,23 Dietary approaches emphasizing hypocaloric, Mediterranean, or anti-inflammatory patterns have demonstrated additional improvements in disease activity, likely through attenuation of systemic inflammation and metabolic dysregulation.21 Bariatric surgery provides more sustained benefits, with multiple studies reporting long-term remission or reduced psoriasis severity following substantial postoperative weight loss.23,24
Pharmacologic weight-loss therapies, particularly glucagonlike peptide-1 receptor agonists, have emerged as potential adjuncts in psoriasis management. Although data remain limited, these agents may reduce systemic inflammation, improve insulin sensitivity, and indirectly enhance biologic response.25
Conclusion
Psoriasis and obesity are interconnected chronic inflammatory conditions that share overlapping cytokine pathways and mutually exacerbate the clinical course. Systemic inflammation driven by cytokines such as TNF-α, IL-6, and IL-17 not only promotes psoriatic skin disease but also contributes to metabolic dysfunction and cardiovascular risk. In turn, excess adiposity amplifies inflammatory signaling and diminishes therapeutic response, creating a self-perpetuating cycle of disease.
Dermatologists should identify obesity-related risks early; counsel patients on lifestyle changes; initiate metabolic screening; and coordinate care across primary care, nutrition, and rheumatology. Regular screening for metabolic syndrome, cardiovascular comorbidities, and psychosocial distress should be integrated into psoriasis management. Future research should focus on personalized treatment strategies that integrate management of inflammatory skin disease with underlying metabolic health, such as optimizing biologic dosing and identifying novel targets that disrupt the pathophysiologic loop. By recognizing and addressing the shared inflammatory mechanisms of psoriasis and obesity, clinicians can improve both dermatologic and systemic outcomes for affected patients.




