Cardiovascular implantable electronic device infection: A stepwise approach to diagnosis and management
ABSTRACTInfection related to cardiovascular implantable electronic devices is a serious complication, necessitating removal of the device and prolonged parenteral antibiotic therapy. Accurate diagnosis and optimal management of these infections are challenging. This review highlights the critical management decisions.
KEY POINTS
- Although inflammatory signs at the generator pocket are the most common presentation of an infection occurring soon after the device is implanted, positive blood cultures may be the sole manifestation of a late-onset endovascular infection.
- Staphylococci are the most common pathogens in both pocket infections and endovascular infections.
- Two sets of blood cultures should be obtained in all patients suspected of having a cardiac device infection.
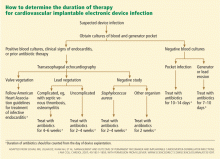
- Transesophageal echocardiography should be ordered in all patients with suspected cardiac device infection who have positive blood cultures, as it can identify intracardiac complications of infection and assess for evidence of cardiac valve involvement.
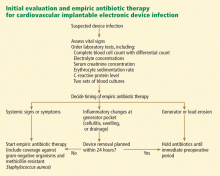
DIAGNOSIS OF INFECTION AND ASSOCIATED COMPLICATIONS
The clinical diagnosis of pocket infection is usually quite straightforward. However, occasionally, an early postoperative pocket hematoma can mimic pocket infection, and distinguishing these two may be difficult. Close collaboration between an internist, cardiologist, and infectious-disease specialist and careful observation of the patient may help to avoid a premature and incorrect diagnosis of pocket infection and unnecessary removal of the device in this scenario.
While diagnosing a pocket infection may be simple, an accurate and timely diagnosis of endovascular infection with an intact pocket can be challenging, especially if echocardiography shows no conclusive evidence of involvement of the device leads. Even when the infection is limited to the generator pocket, attempts to isolate causative pathogens may be hampered if empiric antibiotic therapy is started before culture samples are obtained from the pocket and from the blood.
Complete blood count with differential cell count.
Electrolyte and serum creatinine concentrations.
Inflammatory markers, including erythrocyte sedimentation rate and C-reactive protein concentration.
Swabs for bacterial cultures should be sent if there is purulent drainage from the generator pocket. This can be done in the office before referral to the emergency department or a tertiary care center for inpatient admission. If the pocket appears swollen or fluctuant, needle aspiration should be avoided, as it can introduce organisms and cause contamination.5
Two sets of peripheral blood cultures should be obtained. If the patient has an implanted central venous catheter, blood cultures via each catheter port should also be obtained, as they may help to pinpoint the source of bloodstream infection in cases in which blood culture results are positive.
TEE should also be performed in patients with systemic signs and symptoms (such as fever, chills, malaise, dyspnea, hypotension, or peripheral stigmata of endocarditis) or abnormal test results (leukocytosis, elevated inflammatory markers, or evidence of pulmonary emboli on imaging), even if blood cultures are negative. Similarly, TEE should also be considered in patients in whom blood cultures may be negative as a result of previous antimicrobial therapy.
If a decision is made to remove the device (see below), intraoperative pocket tissue and lead-tip cultures should be sent for Gram staining and bacterial culture. Fungal and mycobacterial cultures may be necessary in immunocompromised hosts, or if Gram staining and bacterial cultures from pocket tissue samples are negative. Caution must be exercised when interpreting the results of lead-tip cultures, as lead tips may become contaminated while being pulled through an infected pocket during removal.20,22
This approach should lead to an accurate diagnosis of CIED-related infection and associated complications in most patients. However, the diagnosis may remain elusive if results of blood cultures are positive but the pocket is intact and there is no echocardiographic evidence of lead or valve involvement. This is especially true in cases of S aureus bacteremia, in which positive blood cultures may be the sole manifestation of underlying device infection.19,23 Factors associated with higher odds of underlying device infection in this scenario include bacteremia lasting more than 24 hours, prosthetic valves, bacteremia within 3 months of device implantation, and no alternative focus of bacteremia.12
Evidence is emerging that underlying device infection should also be considered in patients with bloodstream infection with coagulase-negative staphylococci in the setting of an implanted device.24 On the other hand, seeding of device leads with gram-negative organisms is infrequent, and routine imaging of intracardiac leads is not necessary in cases of gram-negative bacteremia.25
In our opinion, cases of bacteremia in which underlying occult device infection is a concern are best managed by consultation with an infectious disease specialist.