Cervical cancer screening: Less testing, smarter testing
ABSTRACTIn its 2009 recommendations for cervical cancer screening, the American College of Obstetricians and Gynecologists (ACOG) calls for less-frequent but smarter screening that integrates testing for human papillomavirus (HPV) infection with the Papanicolaou (Pap) test. We review the recommendations from this and other organizations and how and why they are evolving.
KEY POINTS
- Persistent infection with one of the 18 high-risk types of HPV is associated with the development of nearly all cases of cervical cancer.
- The 2009 ACOG guidelines recommend starting to screen with the Pap test at an older age (21 years) than in the past, and they recommend a longer screening interval for women in their 20s, ie, every 2 years instead of yearly.
- Women age 30 and older should undergo both Pap and HPV testing. If both tests are negative, screening should be done again no sooner than 3 years. Alternatively, women age 30 or older who have had three consecutive negative Pap tests can be screened by Pap testing every 3 years.
- Although vaccination can prevent most primary infections with high-risk HPV, it does not eliminate the need for continuing cervical cancer screening, as it does not protect against all high-risk HPV subtypes.
- Screening can stop at age 65 to 70 in women who have had three negative Pap tests in a row and no abnormal tests within the past 10 years.
Cervical cancer screening and prevention have evolved rapidly in the last decade, especially in the 5 years since the introduction of the first cancer prevention vaccine, human papillomavirus (HPV) recombinant vaccine.1
Providers need to understand the rationale for the recommendations so that they can explain them to patients. In particular, patients may wonder why we now begin screening for cervical cancer later than we used to, and why some women do not need to be screened as often. Both of these changes result from our enhanced understanding of the role of HPV in cervical cancer genesis.
In this article we will briefly review:
- The current understanding of the natural history of cervical cancer
- Advantages and disadvantages of cervical cytology, ie, the Papanicolaou (Pap) test
- The role of HPV testing in cervical cancer screening
- The latest screening guidelines (the new standard of care)
- A possible future screening strategy
- The impact of HPV vaccination on screening.
500,000 NEW CASES EVERY YEAR
The incidence of cervical cancer and its mortality rate have decreased more than 50% in the United States over the past 3 decades, largely as a result of screening with the Pap test.2 In 2010, there were an estimated 12,200 new cases of invasive cervical cancer in the United States and 4,210 deaths from it,3 which are lower than the historical rates.
However, because most developing countries lack effective screening programs, cervical cancer remains the second-leading cause of death from cancer in women worldwide. According to a recent estimate, there are almost 500,000 new cases and 240,000 deaths from this disease worldwide every year.4 If effective global screening programs could be set up, they would markedly reduce the incidence of cervical cancer and deaths from it.5
HPV IS NECESSARY BUT NOT SUFFICIENT FOR CERVICAL CANCER TO DEVELOP
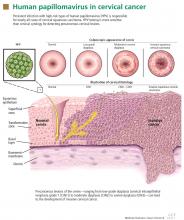
For cervical cancer to develop, the essential first step is infection of the cervical epithelium with one of the oncogenic (high-risk) types of HPV (see below).6–10 Walboomers et al9 tested cervical tissue samples taken from 932 women with cervical cancer and detected HPV DNA in 930 (99.8%) of them.
Fortunately, most HPV-infected women do not develop cervical cancer, as most young women clear the infection in an average of 8 to 24 months.11,12 However, if the infection persists, and if it is one of the high-risk types of HPV, precursor lesions can develop that can progress to cervical cancer.13 The evidence conclusively supports the association between oncogenic HPV infection and the subsequent development of virtually all cases of cervical cancer.6–10
Known risk factors for HPV persistence and the subsequent development of high-grade lesions are cigarette smoking and a compromised immune system.14,15
Terminology: Results of Pap smears
- Normal
- Atypical squamous cells of undetermined significance (ASC-US)
- Low-grade squamous intraepithelial lesions (LSIL)
- High-grade squamous intraepithelial lesions (HSIL)
- Cancer.
Terminology: Results of cervical biopsy
- Normal
- Cervical intraepithelial neoplasia grade 1 (CIN1)
- CIN2 (previously called moderate dysplasia)
- CIN3 (previously called severe dysplasia)
- Carcinoma in situ
- Invasive cervical cancer.
Lesions that are CIN2 or higher are considered high-grade.13
The 18 high-risk HPV types
More than 40 types of HPV infect the genital tract16; 18 of these (types 16, 18, 26, 31, 33, 35, 39, 45, 51, 52, 53, 56, 58, 59, 66, 68, 73, and 82) are classified as high-risk because of their causative association with cervical cancer (ie, their oncogenic potential).17
How HPV causes cervical cancer
In laboratory cultures, normal human cells die out after a few generations. However, if human epithelial cells are infected by one of the high-risk types of HPV, they can go on dividing indefinitely.18,19
Two HPV proteins, E6 and E7, induce this cell “immortalization.”20,21 E6 from high-risk HPV binds to the human tumor-suppressor protein p53 and rapidly degrades it in a proteolytic process. The p53 protein normally suppresses cell proliferation by arresting growth in the G1 phase of the cell cycle. Therefore, with less p53, the cell cannot suppress uncontrolled cell growth.22–24
Similarly, E7 from high-risk HPV forms a complex with another human tumor suppressor, the retinoblastoma protein (pRB), and disrupts its binding to a transcriptional factor, E2F-1. The freed E2F-1 then stimulates DNA synthesis and uncontrolled cell growth.25
Furthermore, HPV-16 E6 and E7 proteins can collectively cause cellular genetic instability.26
The carcinogenic mechanism of high-risk HPV is complex. The host immune system and natural tumor suppression play important roles. However, the natural history of cervical intraepithelial neoplasia is not well understood. For example, it remains unclear if low-grade lesions such as CIN1 are necessary precursors to high-grade lesions and invasive cancer.6,7,10