Bone density vs bone quality: What’s a clinician to do?
ABSTRACTStudies of the epidemiology of osteoporosis and of drug treatments for it have challenged the concept that denser bone means stronger bone. Bone strength or resistance to fracture is not easily measured by routine densitometry, being a function of both density and quality.
KEY POINTS
- Bone quality is a composite of properties that make bone resist fracture, such as its microarchitecture, accumulated microscopic damage, the quality of collagen, mineral crystal size, and bone turnover.
- The T score was derived from a population of white women in their mid to late 60s and older; in other populations, low T scores do not necessarily reflect the disease state—osteoporosis—with its inherent decreased strength and propensity to fracture.
- In assessing the risk of fractures, clinicians should consider not only the bone mineral density but also clinical risk factors.
- Markers of bone turnover are elevated in some cases of primary osteoporosis and return to normal levels with antiresorptive therapy but not with anabolic therapy.
Most clinicians were taught directly or indirectly that bone density is the gauge for assessing bone strength and the response to antiosteoporotic treatment. In recent years, however, the concept of bone strength has moved beyond density alone and has expanded to include a number of characteristics of bone that collectively are called quality.
This paper describes how the notion of quality has emerged and some of the clinical scenarios in which quality applies. It discusses several observations in the clinical literature that challenge our understanding of bone density and strength and provides the practitioner a better understanding of densitometry in clinical practice.
WHAT IS BONE QUALITY?
Bone quality is not precisely defined. It is described operationally as an amalgamation of all the factors that determine how well the skeleton can resist fracturing, such as microarchitecture, accumulated microscopic damage, the quality of collagen, the size of mineral crystals, and the rate of bone turnover. The term became popular in the early 1990s, when paradoxes in the treatment of osteoporosis challenged the generally accepted orthodoxy that bone density itself was the best way to assess strength of bone.
FROM BONE MASS TO T SCORES TO BONE QUALITY
Today’s practitioners appreciate the importance of the T score in diagnosing osteoporosis. It was not always this way, since the early attempts to use bone densitometry focused on a specific cutoff of bone mass as a risk for fracture and not the statistical T scores or Z scores that we know.1–3
The T score concept was originally developed to assess the probability of fragility fractures in postmenopausal white women in their mid to late 60s and older.4 It has been useful because the disease prevalence is high in this age group. The T score as originally used was a surrogate marker for the histologic changes in aged bone that render it weak and susceptible to fractures from low loading forces: the lower the score, the worse the fracture risk. It followed intuitively that a low T score clinched the diagnosis of primary osteoporosis.
But the T score has its problems when used outside this intended population. Practitioners have assumed that all patients with abnormally low scores have primary osteoporosis. However, this number alone is insufficient to accurately make such a diagnosis in patients outside the demographic group in which it was developed, because the low disease prevalence in younger groups makes the score less accurate as a predictive tool. Moreover, reevaluation of data from pivotal clinical trials has brought into question our long-held idea that increases in bone density parallel increases in bone strength and reduction in fractures, and that therapeutic improvement in bone density is the mark of success. Bone strength or resistance to fracture is more complex than density alone. Into this arena enters the concept of bone quality, which attempts to explain the following observations.
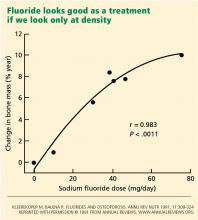
DENSER BONE IS NOT ALWAYS STRONGER
NOT ALL LOW BONE MINERAL DENSITY IS OSTEOPOROSIS
The following case describes a clinical scenario in which a patient has low bone density but does not have osteoporosis.
A young healthy woman with low bone density
A 35-year-old healthy woman who has jogged recreationally for decades is evaluated for possible treatment of osteoporosis. She started to feel back pain after doing heavy work in her garden. Spinal radiographs did not show a reason for her pain, but her physician, concerned about osteopenia, sent her for dual-energy x-ray absorptiometry. Her spinal T scores and Z scores were 2.5 standard deviations below the mean.
Should she start pharmacologic therapy?
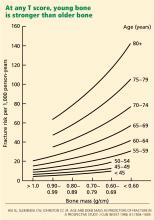
Young bone is stronger than older bone
This case shows the other end of the spectrum from the fluoride story. Here, a young healthy person inappropriately underwent a density scan, which led to confusion about how to interpret the results.
As stated above, T scores are not appropriate for young patients—the Z score is used instead. In this case, the low value implied deficiency of bone mass compared with age-matched norms. However, in this patient with no clinical risk factors for fracture, a low T score meant that her bone density was low, but not that she had osteoporosis.
Several factors could account for her low bone density. It could be genetic, if her family is small in stature, or she could be at the extreme end of the distribution curve for normal individuals. Runners tend to be slight in build, and so may have lighter bones. Furthermore, for women, excessive running could lead to lower estrogen activity and therefore lower bone mineral density.
Drug treatment is not warranted for this patient, but standard therapy with exercise, vitamin D, and adequate elemental calcium from the diet or supplements is reasonable.
Thus, the notion of quality entered the clinical arena. Young bone and older bone are qualitatively different in strength, even with similar bone density. This difference was later found to be related to significant qualitative changes within the microscopic architecture of the bone, the collagen, the mineral, and the physiologic activity of the skeletal cells—elements that the T score does not reflect.
Hence, young bone is stronger than older bone across all levels of bone mass or T scores. Its quality is better.