Skin manifestations of diabetes
ABSTRACTDiabetes mellitus can be complicated by a variety of cutaneous manifestations. Good metabolic control may prevent some of these manifestations and may support cure. Unfortunately, most glucose-lowering drugs also have cutaneous side effects. It is important to be able to recognize these signs and symptoms and to either treat them appropriately or refer the patient to a dermatologist or diabetologist.
KEY POINTS
- Patients with type 2 diabetes more often develop skin infections, whereas those with type 1 more often have autoimmune-related lesions.
- Insulin signaling supports normal skin proliferation, differentiation, and maintenance, and a lack of insulin may lead to impaired wound healing, which may affect insulin resorption.
- Skin manifestations of diabetes may also serve as ports of entry for secondary infection.
- A candidal infection (moniliasis) can be an early sign of undiagnosed diabetes.
- Watch for dermal side effects of insulin injections and oral hypoglycemic drugs.
MANIFESTATIONS ASSOCIATED WITH TYPE 1 DIABETES
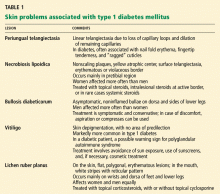
Periungual telangiectasia
The lesions of periungual telangiectasia, appearing as red, dilated, capillary veins, are easily visible with the naked eye and are the result of a loss of capillary loops and dilation of the remaining capillaries. A prevalence up to 49% has been described in all diabetic patients.1 Connective tissue diseases may also involve periungual telangiectases, although these lesions are morphologically different. In diabetes, periungual telangiectasia is often associated with nail fold erythema, accompanied by fingertip tenderness and “ragged” cuticles.2
Necrobiosis lipoidica
Metabolic control has no proven effect on the course of this condition,6 although Cohen et al7 reported that tight glucose control reduced the incidence in diabetic patients. Treatment includes application of a topical steroid with or without occlusion; intralesional steroids at the active border; or, in the rare severe or extensive case, systemic steroids.6,7 In some resistant cases, aspirin, chloroquine (Aralen), and cyclosporine (Sandimmune, Neoral) have been used with some success.3,8,9
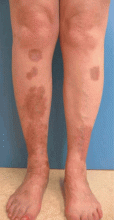
Bullosis diabeticorum
Bullosis diabeticorum develops in approximately 0.5% of diabetic patients, but more often in those with type 1 diabetes, and more often in men and in patients with long-standing diabetes with peripheral neuropathy. It presents as asymptomatic bullae containing sterile fluid on a noninflamed base, usually arising spontaneously on the dorsa and sides of the lower legs and feet, sometimes on the hands or the forearms. The cause is unknown, and it is a diagnosis of exclusion. The differential diagnosis includes epidermolysis bullosa acquisita, porphyria cutanea tarda, bullous pemphigoid, bullous impetigo, coma blisters, and erythema multiforme.
Treatment is symptomatic and conservative. In case of discomfort, the bullae can be aspirated (leaving the blister roof intact), or compresses can be used. Topical antibiotics may be required to prevent secondary infection.3 Most lesions resolve in 2 to 3 weeks without residual scarring.5,6
Vitiligo
Vitiligo vulgaris, or skin depigmentation, occurs more often in type 1 diabetic patients. From 1% to 7% of all diabetic patients have vitiligo vs 0.2% to 1% of the general population. The mechanism behind the association has not been elucidated, although some have suggested polyglandular autoimmune syndrome (PAS), a rare immune endocrinopathy characterized by the coexistence of at least two endocrine gland insufficiencies that are based on autoimmune mechanisms. PAS type 2 is more common (estimated prevalence of 1:20,000), occurs mainly in the third or fourth decade, and is characterized by adrenal failure, autoimmune thyroid disease, or type 1 diabetes. Adrenal failure may precede other endocrinopathies. Vitiligo and gonadal failure occur more frequently in PAS type 1 than in PAS type 2, whereas immunogastritis, pernicious anemia, and alopecia areata are the main features of PAS type 2. In contrast to PAS type 1, family members of PAS type 2 patients are often affected as well. PAS type 2 is believed to be polygenic, with an autosomal dominant pattern of inheritance.10
Treatment of vitiligo is unsatisfactory in general. Patients should be advised to avoid the sun and to use broad-spectrum sunscreens. For localized vitiligo, topical corticosteroids are preferred, whereas for generalized vitiligo ultraviolet B light treatment is most effective. Cosmetic treatment is an option for improved well-being.11
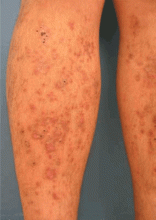
Oral lichen planus
Clinically, lichen planus presents as polygonal erythematous flat lesions. Most often affected are the wrists, the dorsa of the feet, and the lower legs. Oral lichen planus presents as white stripes in a reticular pattern.
Clinical and histopathologic differentiation of these lesions from lichenoid reactions to drugs (eg, nonsteroidal anti-inflammatory drugs, antihypertensive drugs) may be difficult, although numerous eosinophils, parakeratosis, and perivascular inflammation around the mid and deep dermal plexuses, are seen in lichenoid drug reactions, but generally not in lichen planus.13
Treatment consists of topical corticosteroids or topical cyclosporine, or both.6