Perioperative management of anemia: Limits of blood transfusion and alternatives to it
ABSTRACT
Perioperative anemia is associated with excess morbidity and mortality. Transfusion of allogeneic blood has been a long-standing strategy for managing perioperative anemia, but the blood supply is insufficient to meet transfusion needs, and complications such as infection, renal injury, and acute lung injury are fairly common. Further, data suggest that mortality and length of stay are worsened with liberal use of transfusion. Medical alternatives to transfusion include iron supplementation and erythropoiesis-stimulating agents (ESAs). Though ESAs reduce the need for perioperative blood transfusion compared with placebo, they are associated with an increased risk of thrombotic events in surgical patients. Cleveland Clinic has been developing a blood management program aimed at reducing allogeneic blood exposure for greater patient safety; the program has achieved some reduction in blood utilization in its first 7 months.
KEY POINTS
- Anemia is a potent multiplier of morbidity and mortality risk, including in the perioperative setting.
- The Joint Commission plans to implement a performance measure on blood management in the near future.
- While the safety of the blood supply has improved markedly from the standpoint of infection transmission, other risks from transfusion persist, including transfusion-related acute lung injury and emerging infections.
- The preoperative evaluation should elicit a history of bleeding tendencies, previous transfusions, and symptoms of anemia. Medications should be reviewed with an eye toward those that may need to be stopped to avoid a predisposition to bleeding (eg, antiplatelets, anticoagulants).
- Use of ESAs minimizes the need for blood transfusion in patients undergoing orthopedic and other surgeries, but they raise the risk of thromboembolism in the absence of prophylactic anticoagulation.
Outcomes and duration of blood storage
An interesting factor in the relation between transfusion and outcomes is the shelf life of the blood being transfused. The FDA currently allows storage of blood for a maximum of 42 days, but a recent study of patients who received red blood cell transfusions during cardiac surgery found that those who received “older blood” (stored for > 14 days) had significantly higher rates of sepsis, prolonged intubation, renal failure, in-hospital mortality, and 1-year mortality compared with those who received “newer blood” (stored for ≤ 14 days).24
These differing outcomes are generally attributed to the so-called storage defect: as blood gets older, it loses components such as 2,3-DPG and adenosine disphosphate, its red cells lose deformability, and it undergoes buildup of cytokines and free hemoglobin. Increased demand for newer blood in light of the storage defect could further intensify pressures on the blood supply.
MANAGEMENT OF PERIOPERATIVE ANEMIA
In light of these shortcomings of blood transfusion, how should anemia be managed perioperatively to reduce or avoid the need for transfusion?
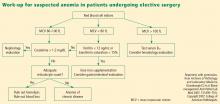
Preoperative evaluation
Vigilance for anemia and related issues in the preoperative evaluation is fundamental. The evaluation should elicit a history of bleeding tendencies, previous transfusions, and symptoms of anemia. Medications should be reviewed with an eye toward any that may predispose to perioperative bleeding and anemia, such as aspirin, clopidogrel, and anticoagulants. During the physical examination, alertness for pallor and petechiae is key, as is attentiveness to symptoms of anemia such as shortness of breath and fatigue.
Overview of management options
Once the cause of anemia is identified, the choice for optimal medical management can be made. Choices broadly consist of pharmacologic and technological options. The former include iron supplements and erythropoiesis-stimulating agents. Among other pharmacologic options are thrombin, collagen, fibrin glue, tranexamic acid, and aminocaproic acid, but these agents are less well studied and will not be discussed here. Technological options include preoperative autologous blood donation, cell salvage, and acute normovolemic hemodilution.
In addition to these options, careful management of anticoagulant and antiplatelet medications should be provided, including discontinuation or substitution of drugs that could hamper clotting perioperatively.
PHARMACOLOGIC OPTIONS
Iron supplementation
Oral iron is available in four preparations: ferrous sulfate, ferrous gluconate, ferrous fumarate, and iron polysaccharide. Gastrointestinal side effects may limit these preparations’ tolerability. Iron supplements with a high elemental value will require fewer pills and fewer doses, reducing the risk or frequency of side effects.
Intravenous (IV) iron preparations are much safer now than they were years ago, when anaphylactic reactions were a concern. The ones generally used in the perioperative setting are iron sucrose and iron gluconate. Unlike the older IV preparations, the use of iron sucrose and iron gluconate often requires a second dose. The effect on hemoglobin levels usually occurs starting at 1 week, with the maximum effect achieved at 2 weeks. Hypotension, arthralgia, abdominal discomfort, and back pain are potential side effects of IV iron.
Efficacy and safety of iron supplementation. Evidence of the efficacy of preoperative iron supplementation is mounting. A study of 569 patients undergoing colorectal cancer surgery found that among the 116 patients who were anemic, intraoperative transfusion was needed in a significantly lower proportion of those who received 2 weeks of preoperative oral iron supplementation (200 mg) compared with those who received no iron therapy (9.4% vs 27.4%; P < .05).26 Similarly, in an uncontrolled study, 10 days of IV iron sucrose starting 4 weeks preoperatively significantly increased hemoglobin levels in 20 patients with iron-deficiency anemia prior to elective orthopedic surgery.27
Risks of infection and cancer progression have been concerns with IV iron therapy. However, no significant association between IV iron therapy and bacteremia was identified in a prospective study of 985 patients receiving chronic hemodialysis.28 The effect of IV iron administration on tumor progression has not been prospectively studied.
In general, IV iron, especially the newer forms, is a safer alternative to blood transfusion. Death occurs at a much lower rate with iron than with blood transfusion (0.4 per million vs 4 per million, respectively), as do life-threatening adverse events (4 per million vs 10 per million, respectively), according to a systematic review by the Network for Advancement of Transfusion Alternatives.29
Erythropoiesis-stimulating agents
Erythropoiesis-stimulating agents (ESAs) include epoetin alfa (erythropoietin), first approved by the FDA in 1989, and the more recently introduced darbepoetin alfa. They are approved to treat anemia in several patient populations, but only epoetin alfa is approved by the FDA explicitly for use in patients undergoing major surgery (to reduce the need for blood transfusions). The ESAs have come under intense scrutiny in recent years over their risk-to-benefit ratio, as detailed below.
The preoperative dosing schedule for epoetin alfa is usually three weekly doses (plus a fourth dose on the day of surgery) if the surgery is scheduled 3 or more weeks in advance. However, daily dosing can be used effectively if the preoperative period is less than 3 weeks, provided that it is continued until 4 days after surgery. Oral iron is necessary throughout the course of epoetin alfa therapy.
Efficacy in reducing transfusions. In a systematic review published in 1998, epoetin alfa was shown to minimize perioperative exposure to allogeneic blood transfusion in patients undergoing orthopedic or cardiac surgery.30 Its benefit was greatest in patients at the highest risk of requiring transfusion. It was effective whether given daily or weekly, and did not significantly increase the risk of thrombotic events when used in surgical patients, although some studies did find an excess of thrombotic events with its use.
In three randomized trials conducted in patients undergoing joint arthroplasty (hip or knee), epoetin alfa was associated with substantial and significant reductions in perioperative blood transfusion compared with placebo or preoperative autologous blood donation.31–33 Rates of deep vein thrombosis (DVT) did not differ significantly between the epoetin alfa and placebo groups.
Concerns over perioperative thromboembolic risk. In early 2007, the FDA was made aware of preliminary results of an open-label study in which 681 patients undergoing elective spinal surgery who did not receive prophylactic anticoagulation were randomized to epoetin alfa plus standard-of-care therapy (pneumatic compression) or standard-of-care therapy alone.34,35 The incidence of DVT was 4.7% in patients treated with epoetin alfa compared with 2.1% in those not receiving epoetin alfa. It is important to note that the available ESAs are prothrombotic and increase thrombotic risk significantly, especially in populations like this one in which pharmacologic DVT prophylaxis is not routinely used.
Based in part on this study, the FDA in 2007 required a boxed warning to be added to the ESAs’ package inserts to specify the increased risk of DVT with their use in surgical patients not receiving prophylactic anticoagulation. The warning urges consideration of the use of DVT prophylaxis in surgical patients receiving an ESA.34,35