Update on the management of venous thromboembolism
ABSTRACT
Venous thromboembolism (VTE), which includes deep vein thrombosis (DVT) and pulmonary embolism, is a common cardiovascular disease associated with significant morbidity ranging from painful leg swelling, chest pain, shortness of breath, and even death. Long-term complications include recurrent VTE, postpulmonary embolism syndrome, chronic thromboembolic pulmonary hypertension, and postthrombotic syndrome (PTS). Management of VTE requires immediate anticoagulation therapy based on a risk assessment for bleeding. Direct oral anticoagulants (DOACs) have become an important option for patients as reflected in the most recent American College of Chest Physician treatment guidelines.
KEY POINTS
- VTE treatment should begin immediately with heparin, low-molecular-weight heparin (LMWH), fondaparinux, or the DOACs (rivaroxaban or apixaban) in patients deemed appropriate based on a risk assessment for bleeding.
- For patients with VTE and no cancer, long-term treatment with dabigatran, rivaroxaban, apixaban, or edoxaban is recommended over the vitamin K antagonists (VKA).
- LMWH is recommended for the long-term treatment of VTE in patients with cancer.
- For extended-duration anticoagulation, the DOACs (dabigatran, rivaroxaban and apixaban) and the VKA antagonists are options.
- Compression stockings are no longer recommended for prevention of PTS in patients with acute DVT but may be beneficial symptomatically.
Venous thromboembolism (VTE) includes both deep vein thrombosis (DVT) and pulmonary embolism (PE). Although the exact incidence of VTE is unknown, an estimated 1 million people in the United States are affected each year, with about a third experiencing a recurrence within 10 years.1 VTE affects hospitalized and nonhospitalized patients, is often overlooked, and results in long-term complications including postthrombotic syndrome (PTS) for DVT, postpulmonary embolism syndrome and chronic thromboembolic pulmonary hypertension for PE, and death.2
TREATMENT
Treatment for VTE should be initiated in the following cases:
- Proximal DVT of the lower extremity
- Symptomatic distal (calf vein) DVT
- Symptomatic upper extremity DVT (axillary-subclavian veins)
- PE
- Subsegmental PE in a patient at risk for recurrence
- Surveillance for subsegmental PE in a patient with no proximal DVT and a low risk of recurrence.
In addition to anticoagulants, other more aggressive therapies for VTE may be appropriate, such as systemic thrombolysis in the case of PE or catheter-directed thrombolytic or pharmacomechnical therapies for DVT or PE, surgical intervention (acute pulmonary embolectomy), or placement of an inferior vena cava (IVC) filter.
This article reviews the management of VTE, highlighting the recent changes in treatment and prevention guidelines from the American College of Chest Physicians (ACCP).3
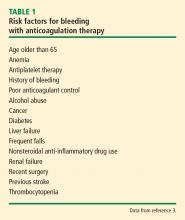
Risk of bleeding
In assessing a patient’s risk of bleeding for anticoagulation therapy (Table 1), the absence of risk factors is considered low risk for bleeding, the presence of 1 risk factor is considered intermediate risk, and 2 or more risk factors is considered high risk. Compared with low-risk patients, moderate-risk patients have a twofold increased risk of major bleeding and high-risk patients have an eightfold increased risk of major bleeding. This equates to an annualized risk of major bleeding of 0.8% for low-risk patients, 1.6% for moderate-risk patients, and greater than 6.5% for high-risk patients.3
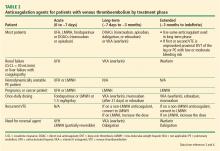
Anticoagulants
Deciding on which anticoagulant to use depends on the indication, the patient’s underlying condition, the patient’s preference, and the patient’s risk of bleeding. Heparin, the LMWHs, fondaparinux and the DOACs (rivaroxaban and apixaban) are the only agents approved by the US Food and Drug Administration (FDA) recommended for the acute treatment phase, while the DOACs and warfarin are anticoagulation options for the long-term and extended treatment phases. The LMWHs should be used for the patient with cancer and during pregnancy.
Unfractionated heparin. UFH is administered parenterally and can be used for the prevention and treatment of VTE. Heparin remains an option for initial treatment of patients with acute VTE and is generally preferred over LMWH for patients who may require advanced therapies, such as for hemodynamically unstable PE or iliofemoral DVT. It is also recommended for patients with renal failure.3 Weight-based dosing (80 U/kg bolus followed by 18 U/kg/hour intravenous infusion) is recommended, targeting an antifactor activated clotting factor (anti-Xa) assay level of 0.3 IU/mL to 0.7 IU/mL. Heparin may also be given subcutaneously in an outpatient setting using an initial bolus of 333 U/kg followed by a subcutaneous dose of 17,500 U twice daily.5
Low-molecular-weight heparin. LMWHs are administered as weight-based subcutaneous injections and have indications for patients with acute VTE and for VTE prophylaxis. LMWHs are used for transitioning to warfarin, dabigatran, or edoxaban for long-term anticoagulation and are recommended over warfarin and DOACs for treatment of VTE in patients with cancer and in pregnant women.3
Enoxaparin (Lovenox), the most commonly used agent in the United States, is given either as a once-daily injection (1.5 mg/kg/day) or a twice-daily injection (1 mg/kg every 12 hours). It is also approved for VTE prophylaxis in patients undergoing hip or knee replacement surgery or abdominal surgery, or in patients with severely restricted mobility during acute illness. LMWH can also be given in patients with renal insufficiency (creatinine clearance [CrCL] < 30 mL/minute) after dose adjustment. No monitoring is required, although it is advised in pediatric patients, pregnant women, obese patients, and patients with renal insufficiency. If monitoring is required, an anti-Xa assay using LMWH as a reference standard should be done 4 hours after subcutaneous injection. The therapeutic range for enoxaparin is 0.5 IU/mL to 1.0 IU/mL for the 12-hour regimen and greater than 1.0 IU/mL for the once-daily dose. Other LMWHs available in the United States include dalteparin (Fragmin) and tinzaparin (Innohep). Each has its own specific indications.
Fondaparinux. Fondaparinux is an indirect factor Xa inhibitor, chemically related to LMWH. It is approved for treatment of patients with acute VTE when used in combination with a VKA (warfarin) or dabigatran or edoxaban. It also has approval for VTE prophylaxis in patients undergoing hip fracture, hip or knee replacement, and abdominal surgery. Fondaparinux is administered as a once-daily subcutaneous injection of 2.5 mg for DVT prophylaxis and a body weight-based dose for the treatment of VTE (5 mg < 50 kg; 7.5 mg 50 to 100 kg; 10 mg > 100 kg).6 Fondaparinux is contraindicated in patients with severe renal impairment (CrCL les 30 mL/min) and bacterial endocarditis.6
Warfarin. Warfarin, a VKA, was the mainstay of therapy for long-term and extended treatment of VTE until the advent of the DOACs. Warfarin must be coadministered with heparin, LMWH, or fondaparinux initially and continued as overlap therapy for a minimum of 5 days until the international normalized ratio [INR] is at least 2.0 for 24 hours.4 Early initiation of a VKA on the first day of parenteral therapy is advised.
Warfarin remains the best option for patients on long-term or extended anticoagulation with liver dysfunction (elevated serum transaminases exceeding twice the upper limits of normal or active liver disease) or renal disease (CrCL < 30 mL/min), as well as patients unable to afford DOACs. Additionally, select patient populations may still be best served by warfarin as these groups were underrepresented or not included in DOAC trials, including pediatric patients, individuals with body weight less than 50 kg or greater than 150 kg, and patients with select types of thrombophilia (eg, antiphospholipid syndrome). Warfarin is also advised for patients with poor compliance, as international normalized ratio of prothrombin time (PT/INR) monitoring is required using a point-of-care testing device or during a visit to an anticoagulation clinic. DOACs do not require monitoring, and noncompliance will not be readily apparent.