PCSK9 inhibition: A promise fulfilled?
ABSTRACTThe association of reduced proprotein convertase subtilisin/kexin type 9 (PCSK9) activity with reduced cardiovascular disease (CVD) events—and the need for add-ons to statin therapy to achieve treatment goals—has led to the rapid development and US Food and Drug Administration (FDA) approval of monoclonal antibody therapies to inhibit PCSK9. Now that PCSK9 inhibitors are approved by the FDA for use in certain patients, data from ongoing long-term clinical trials addressing tolerability, safety, and proof of additional reduction in CVD events are eagerly awaited
KEY POINTS
- Potential candidates for PCSK9 inhibitor therapy are patients with familial hypercholesterolemia with a lifetime burden of elevated low-density-lipoprotein cholesterol (LDL-C) and thus a low likelihood of optimal control on current therapies; patients with complete or partial statin intolerance, with high-intensity statin dosing limited by adverse effects; and patients at high CVD risk with LDL-C goals not achieved with current therapies.
- Subcutaneously administered monoclonal antibodies targeting PCSK9 are currently the only PCSK9 inhibitors with FDA approval.
- PCSK9 inhibitors under study include agents with more durable effect and that require less frequent injections, RNA-interference therapies, vaccinations, antisense therapies, and oral formulations.
PCSK9 INHIBITORS: CLINICAL TRIALS
Subcutaneously administered monoclonal antibodies targeting PCSK9 currently are the only PCSK9 inhibitors FDA-approved for clinical use. The first study to demonstrate efficacy in enhancing uptake of serum LDL-C was performed in 2009.33 Multiple phase 1 and 2 studies soon followed, demonstrating acceptable safety and 50% to 70% reductions in LDL-C at upper-dose titrations.34 Additionally, there were significant reductions in total cholesterol, ApoB, triglycerides, and lipoprotein(a).
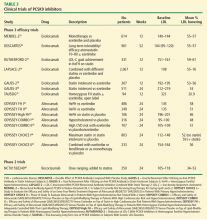
These early developments paved the way for larger phase 3 trials (Table 3).35–48 The PCSK9 inhibitors evolocumab and alirocumab have been shown in multiple phase 3 clinical trials to achieve a consistent dose-dependent 50% to 60% reduction in LDL-C across a broad range of CVD risk, pretreatment LDL-C levels, and background therapy: monotherapy (MENDEL-2, ODYSSEY COMBO I),35,44 added to statin therapy (LAPLACE-2, ODYSSEY CHOICE I),38,46 and in individuals with heterozygous FH (RUTHERFORD-2, ODYSSEY-FH).37,42 Trials with bococizumab are under way.
The GAUSS-2 clinical trial (Goal Achievement after Utilizing an Anti-PCSK9 Antibody in Statin Intolerant Subjects-2) demonstrated similar efficacy in reducing LDL-C in patients with clinically assessed statin intolerance due to muscle-related adverse symptoms.39 In GAUSS-3, patients were first identified as being statin intolerant secondary to muscle-associated symptoms based on a randomized, crossover trial of atorvastatin vs placebo.40 The 43% of participants who experienced intolerable muscle-related symptoms on the statin but not on placebo were then randomized to evolocumab vs ezetimibe. Results showed significant reduction in LDL-C in the evolocumab group (52.8%) compared with the ezetimibe group (16.7%). Additionally, among patients with muscle symptoms on statin therapy, PCSK9 therapy was discontinued for muscle symptoms in only 0.7% of evolocumab recipients and 6.8% of ezetimibe recipients.
Overall, the PCSK9 inhibitors are generally well tolerated with injection site reactions being the most common side effect. A meta-analysis published in 2015 of 25 trials including more than 12,000 patients treated with evolocumab and alirocumab reported no significant difference in adverse events or safety outcomes vs placebo or ezetimibe.49 Antidrug binding or neutralizing antibody production to these agents, thus far, has not been shown to be an issue. Additional analyses have not indicated an adverse effect on gonadal hormone levels or increased incidence of new-onset diabetes.
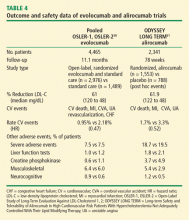
Two studies published in 2015 offer insight into longer term durability and safety as well as potential CVD outcome benefit (Table 4)50,51:
- OSLER-1 and 2: Open-Label Study of Long-Term Evaluation against LDL-Cholesterol (OSLER) trials—evolocumab trial;50
- ODYSSEY long term: Long-Term Safety and Tolerability of Alirocumab in High Cardiovascular Risk Patients with Hypercholesterolemia Not Adequately Controlled with Their Lipid Modifying Therapy—alirocumab trial.51
The OSLER trials reported durable LDL-C reductions of 61% and the ODYSSEY trial reported a LDL-C reduction of 62%.50,51 In both studies, the overall occurrence of adverse events was similar to placebo, but both reported a higher rate of neurocognitive effects in the active treatment groups (evolocumab 0.9% vs 0.3% for standard therapy; alirocumab 1.2% vs 0.5% for placebo). It must be noted that although the absolute rate of neurocognitive adverse events is low, it is unclear if these events were related to the drugs themselves or to extreme lowering of LDL-C. Nevertheless, the FDA has raised concerns about neurocognitive events. A sub-study of the ongoing FOURIER trial with evolocumab—EBBINGHAUS—is expected to address this concern.
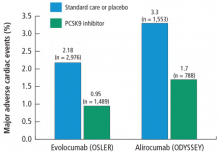
In addition, analyses of CV events showed that the PCSK9 inhibitors effectively cut the CV rate in half in both studies (Figure 1).50,51 In the OSLER trials,50 evolocumab recipients had 53% reduction in major CV events (0.95% vs 2.18% in the standard therapy group; P = .003). In ODYSSEY,51 alirocumab recipients had a 48% reduction in major CV events (1.7% vs 3.3% for placebo; P = .02). Furthermore, a 2015 meta-analysis of 24 phase 2 and 3 trials reported a statistically significant 55% reduction in all-cause mortality and 50% reduction in CV mortality with PCSK9 inhibitors.52
For many reasons including short length of follow-up, study design, and small numbers of outcome events, the OSLER and ODYSSEY studies, although enticing, are exploratory and hypothesis-generating only and results need to be interpreted with caution. Nevertheless, they have set the stage for ongoing prospective randomized outcome trials that are studying the CV effects and tolerability of PCSK9 inhibitors over a longer time frame. These include the following trials.
- The Further Cardiovascular Outcomes Research with PCSK9 Inhibition in Subjects With Elevated Risk (FOURIER) is an ongoing trial with the primary end point of CV death, MI, hospitalization for unstable angina, stroke, or coronary revascularization in high-risk patients receiving evolocumab or placebo.53
- The ODYSSEY trial is examining the effect of alirocumab vs placebo on the composite primary endpoint of coronary heart disease death, non-fatal MI, fatal and nonfatal ischemic stroke, and unstable angina requiring hospitalization in patients who have had an acute coronary syndrome event during the previous 4 to 52 weeks.54
- The Evaluation of Bococizumab in Reducing the Occurrence of Major Cardiovascular Events in High Risk Subjects (SPIRE) trials are investigating the effect of bococizumab, a third PCSK9 “humanized” monoclonal antibody, vs placebo in reducing death, MI, stroke, or unstable angina in patients at high-risk of CVD who are receiving standard lipid-lowering therapy with LDL-C > 70 mg/dL (1.8 mmol/L) (SPIRE-1) or > 100 mg/dL (2.6 mmol/L) (SPIRE-2).55,56
Because these outcome trials are attempting to enroll more than 70,000 patients and are event driven, it is difficult to predict when they will be completed (Table 5).53–56 However, recent estimates indicate completion of at least one trial by the end of 2016 or early 2017, with interim analyses of others expected at that time. It is hoped that they will answer the all-important question of whether PCSK9 inhibitors are associated with further CV event reduction benefit.
CURRENT FDA INDICATIONS AND GUIDELINES
The two PCSK9 inhibitors approved by the FDA—alirocumab (subcutaneous 75 mg every 2 weeks up titrated to 150 mg) and evolocumab (subcutaneous 140 mg every 2 weeks or 420 mg every 4 weeks)—are both indicated for use with statins in patients with heterozygous FH or known atherosclerotic CVD who require further reduction in LDL-C levels despite lifestyle interventions and use of maximally tolerated statins. Evolocumab has also been approved for use in patients with homozygous FH.
Although PCSK9 inhibitors are not specifically approved for patients unable to tolerate statins, the results of GAUSS-3, which documented that statin intolerance is a real, definable entity and very responsive to PCSK9 inhibition, makes these drugs promising agents for patients intolerant of statins and, thus, unable to benefit from high-intensity stain therapy.
In April 2016, the ACC released a clinical consensus update to their 2013 cholesterol guidelines, which is their first recommendation specifically addressing the use of non-statin therapies, including the newer PCSK9 inhibitors.57 For high-risk patients with clinical atherosclerotic CVD or LDL-C > 190 and failure to achieve at least a 50% reduction in LDL-C on maximally tolerated statin, non-statins may be considered. Ezetimibe, given its safety and tolerability, should be the first additional medication added. Bile acid sequestrants may be used as a second-line therapy if ezetimibe is not tolerated and triglycerides are not elevated. If therapy goals are not met on maximally tolerated statin and ezetimibe, either approved PCSK9 inhibitor can be added or used to replace ezetimibe. The document also specifies that given the lack of long-term safety and efficacy data on the PCSK9 inhibitors, they are not recommended for use in primary prevention patients in the absence of FH.
CONCLUSION
Although statin therapy has been shown to substantially reduce LDL-C and CVD adverse events, there remains a high rate of inadequate goal achievement and residual CVD risk in patients receiving statins. Combination therapies with ezetimibe and resins to further lower LDL-C, fibrates and omega 3 fatty acids to lower triglycerides, and niacin to lower both and raise high-density-liproprotein cholesterol are available, even though additional CV risk reduction is minimal or elusive when these drugs are added to statin therapy.
The link between atherogenic lipoproteins and CVD is strong, and the need to develop therapies in addition to statins to substantially and safely reduce LDL-C remains a priority. The association of reduced PCSK9 activity with reduced LDL-C and CV events has led to rapid development and approval of monoclonal antibody therapies to inhibit PCSK9. In trials, these therapies have shown substantial and durable reductions in LDL-C of more than 50%, with acceptable tolerability. Now that PCSK9 inhibitors are approved by the FDA, extended data about long-term tolerability, safety, and efficacy and, most importantly, demonstration of additional reduction in CVD events are needed. It is hoped that the long-term ongoing trials will provide these data.
For the immediate future, statin therapy will continue to be the cornerstone of lipid and CVD risk management based on their low generic cost, proven CVD risk reduction, and clinicians’ comfort with their use. However, the reliable efficacy of PCSK9 inhibitors and the fact that statin therapy itself increases PCSK9 activity makes the addition of PCSK9 inhibitors to statins an attractive approach in high-risk patients failing to reach LDL-C treatment goals.
Although current indications are limited, there are patients at high CVD risk who would be appropriate candidates for these therapies. These include patients with the following:
- FH with lifetime burden of elevated LDL-C and associated low likelihood of achieving optimal LDL-C control on current available therapies
- Complete or partial statin intolerance with high-intensity statin dosing limited by side effects
- High CV risk who are not at LDL-C goal on current therapies.
Now that the first therapies are available, practitioners can expect newer approaches to tackle PCSK9-mediated LDL-C reduction. Bococizumab is lagging in phase 3 trials, but the SPIRE program is moving forward with special population studies expected to conclude in 2016 and simultaneous long-term outcomes trials. Other PCSK9 inhibitors being investigated include agents with more durable effect requiring less frequent injections, RNA-interference therapies, vaccinations, antisense therapies, and oral formulations.
The PCSK9 inhibitors hold promise as an adjunct to statin therapy. Their eventual clinical role will depend on a balance between substantial LDL-C reductions, long-term safety, tolerability, and reduction in CVD events vs the cost (estimated at $14,000 a year), access from payers, acceptance of injectable therapies, and magnitude of incremental benefit when added to current therapies. Nevertheless, initial clinical trial data are encouraging and these drugs may be an important addition to the therapeutic armamentarium against CVD.