Evolving strategies to prevent stroke and thromboembolism in nonvalvular atrial fibrillation
ABSTRACTStroke prevention in patients with nonvalvular atrial fibrillation relies on an assessment of the individual risks for stroke and bleeding. Patients at high risk for stroke are candidates for anticoagulant therapy. Anticoagulants, however, have substantial bleeding risks that must be weighed in the therapeutic decision. Warfarin has been the traditional choice, but the recently introduced novel oral anticoagulants offer similar efficacy with less bleeding risk. Additionally, they do not require monitoring and have fewer drug interactions and dietary restrictions than warfarin. Several devices, which isolate the left atrial appendage, have become available as treatment options for patients with elevated risks of both thromboembolism and bleeding complications.
KEY POINTS
- Specific risk factor management is as important as anticoagulation when addressing stroke risk.
- The CHADS2 score has been superseded by the CHA2DS2-VASc score, which is more accurate for lower-risk categories.
- Anticoagulant options have increased substantially in the past few years with the introduction of novel oral anticoagulants, including the direct thrombin-inhibitor dabigatran and the factor Xa inhibitors rivaroxaban, apixaban, and edoxaban.
- Most atrial thrombi in patients with nonvalvular atrial fibrillation form in the left atrial appendage (LAA); nonpharmacologic interventions have been developed to block the LAA and reduce the risk of stroke.
Novel oral anticoagulants
Dabigatran. The value of dabigatran for prevention of stroke or thromboembolism in AF was tested in the RE-LY trial (Randomized Evaluation of Long-Term Anticoagulation Therapy).13 In this trial, 18,113 patients with AF at high risk for stroke were randomized to dabigatran (110 mg or 150 mg twice daily) or adjusted-dose warfarin (INR target 2.0–3.0). By intention-to-treat analysis, dabigatran 150 mg was superior to warfarin for stroke prevention. Importantly, the risk of intracranial or life-threatening bleeding was significantly lower for both dabigatran doses compared with warfarin. Of note, gastrointestinal bleeding was more common with dabigatran 150 mg than warfarin; rates were similar for dabigatran 110 mg versus warfarin.
Rivaroxaban. This factor-Xa inhibitor was assessed in ROCKET-AF (Rivaroxaban Once-daily oral direct factor Xa inhibition Compared with vitamin K antagonism for prevention of stroke and Embolism Trial in Atrial Fibrillation).14 In this trial, 14,264 patients with AF and at risk for stroke were randomized to rivaroxaban (20 mg once daily) or warfarin (INR target 2.5). In the warfarin arm, INR was in the therapeutic range only 55% of the time. Results showed rivaroxaban was noninferior, but not superior, to warfarin for the prevention of stroke or systemic thromboembolism, the primary end points. From a safety standpoint, the overall bleeding rates were similar in the treatment arms with less life-threatening (fatal or intracranial) hemorrhage events with rivaroxaban.
Apixaban. This factor-Xa inhibitor was tested for stroke prevention in AF in two separate clinical trials.15,16 In the AVERROES trial (Apixaban Versus Acetylsalicylic Acid to Prevent Strokes),15 5,599 patients with AF deemed “unsuitable” for warfarin were randomized to apixaban (5 mg twice daily) or aspirin (81–324 mg daily). The trial was terminated early due to superiority of apixaban in achieving the primary end point: occurrence of stroke or systemic embolism. Importantly, the risk of major bleeding appeared to be similar with apixaban versus aspirin.
The ARISTOTLE (Apixaban for Reduction in Stroke and Other Thromboembolic Events in Atrial Fibrillation) trial16 randomized 18,201 patients with AF and at least one additional risk factor for stroke to either apixaban 5 mg or warfarin (target INR 2.0–3.0). In this trial, apixaban was superior to warfarin for the prevention of stroke or systemic embolism, the primary efficacy end point. There also appeared to be a mortality benefit with apixaban versus warfarin. Importantly, the risk of major and intracranial bleeding, the primary safety outcomes, occurred at lower rates in the apixaban group.
All three of these oral anticoagulants require dose adjustment in patients with renal insufficiency and are contraindicated in patients with end-stage renal failure. They are not indicated in patients with valvular heart disease or a mechanical heart valves.
NONPHARMACOLOGIC INTERVENTIONS
Most atrial thrombi in patients with nonvalvular AF form in the LAA. Nonpharmacologic interventions have been developed to block the LAA to reduce the risk of stroke. These are especially valuable options for patients who are not candidates for chronic anticoagulation. In patients undergoing mitral valve surgery,17 ligation to close the LAA has become a standard practice at experienced centers. The introduction of less invasive catheter-based interventions to occlude the LAA has provided additional options.
Watchman device
The Watchman implant, a closure device that blocks the LAA, was recently approved by the US Food and Drug Administration (FDA) for stroke prevention in patients with nonvalvular AF who are at increased risk for stroke based on a CHA2DS2-VASc score of 2 or greater; candidates also must have an appropriate rationale for nonpharmacologic therapy. The expandable-cage device is surgically delivered into the LAA (Figure 1), which subsequently endothelializes and isolates the LAA. (Video 1 shows the delivery sheath positioned at the os of the appendage with a confirmatory contrast appendogram. Video 2 shows the delivery of the device into the appendage.) Therapeutic warfarin is required for a minimum of 45 days after implant followed by aspirin and clopidogrel for 6 months and then aspirin alone.
This device was initially tested in the PROTECT AF (Watchman Left Atrial Appendage System for Embolic PROTECTion in Patients with Atrial Fibrillation) trial, a noninferiority trial that randomized patients to either device implant or warfarin.18 Device implant was successful in 91% of patients in whom it was attempted. Overall, the study showed noninferiority of the device to warfarin in terms of the primary efficacy standpoint, which included stroke, systemic embolism, and cardiovascular death; however, this came at the expense of higher incidence of procedure-related complications, which seemed to be dependent on a learning curve with the device.
In the PREVAIL (Evaluation of the Watchman LAA Closure Device in Patients with Atrial Fibrillation Versus Long-Term Warfarin Therapy) trial,19 although noninferiority was not achieved, the event rates were low and the safety of the procedure was much improved. This was also demonstrated in registry data, which showed improved safety with device implantation with increased operator experience.
Of note, both of these trials included only patients who were eligible for warfarin. Another nonrandomized study20 assessed the use of this device in patients with a contraindication to long-term anticoagulation. Results showed a very low incidence of stroke, which was lower than CHADS2-matched controls taking either aspirin or clopidogrel. The FDA has approved this device for stroke prevention in AF.
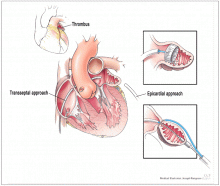
Lariat system
The Lariat system is another percutaneous system for occlusion of the LAA. This device, which requires both atrial transseptal and epicardial access, ligates the appendage from the pericardial space. While some small studies have shown it safe and efficacious in patients with AF who cannot take anticoagulation,21 other reports have not been as encouraging.22 The device has been approved by the FDA for “soft tissue approximation”; it is not approved for stroke prevention.
SUMMARY
The most common serious complications of AF are stroke and thromboembolism. Medical and interventional therapies have been developed to prevent these complications. In patients with an estimated thromboembolic risk of greater than 1% to 2% per year, anticoagulation is warranted to reduce that risk. Warfarin remains one of the most studied and useful medications for this purpose, but its use is limited by the need for frequent monitoring, multiple drug interactions, dietary restrictions, and, most importantly, by the difficulty of consistently maintaining therapeutic INRs.
The recently introduced novel oral anticoagulants have been found to be at least noninferior to warfarin and for some agents to be superior to warfarin for the prevention of stroke and thromboembolism. Their main advantage is that they do not require monitoring and have fewer drug interactions and dietary restrictions. Most important, there appear to be fewer major and life-threatening bleeding events than with warfarin. However, use of these novel agents could be limited by patients’ renal function, which needs to be assessed when these agents are being considered. Another limitation with these agents (versus warfarin) is that patients are not therapeutically anticoagulated when they miss a dose, whereas missing a single dose of warfarin may not have the same effect.
In our practice, we have transitioned to use of these novel oral anticoagulants whenever possible using an approach that assesses the individual patient’s risks for both stroke and thromboembolism as well as the risk for bleeding. In patients who are at increased risk of bleeding but at risk of stroke in AF, percutaneous occlusion of the LAA using the Watchman device is offered. This device has been shown to be noninferior to warfarin for stroke prevention and may provide a survival benefit due to the reduction of life-threatening bleeding, which is an inherent risk with anticoagulants. One caveat is that there is a residual risk of stroke after undergoing LAA occlusion because the same risk factors for stroke in AF contribute to stroke from atherothrombosis and atheroembolism; also, thrombi can form in the body of the left atrium.
Clinical decision-making is often challenging in patients with AF who are at risk of stroke and bleeding. In fact, these risk factors often overlap. In our practice, we have established a multidisciplinary clinic for stroke prevention in AF that involves cardiologists, cardiac electrophysiologists, neurologists, gastroenterologists, and vascular medicine specialists. This model allows a multidisciplinary assessment of patients’ individual risks and ultimately facilitates clinical decision-making in terms of strategies to prevent stroke and thromboembolism in AF.